Absence of molecular evidence of filovirus circulating in bats and rodents in Laikipia North sub-County, Kenya: a cross sectional study
Peris Ambala, Fatima Hussein, Samson Mutura, George Gachara, Nelson Menza, Lindsey McCrickard Shields, Megan Vodzak, Susan Murray, Marc Valitutto, Dawn Zimmerman, Joseph Kamau
Corresponding author: Peris Ambala, Kenyatta University, Biochemistry, Microbiology and Biotechnology Department, Nairobi, Kenya 
Received: 12 Jan 2022 - Accepted: 12 Apr 2022 - Published: 21 Apr 2022
Domain: Epidemiology,Virology
Keywords: Ebola viruses, Marburg virus, filovirus, reservoir, Laikipia, zoonosis
©Peris Ambala et al. PAMJ-One Health (ISSN: 2707-2800). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Peris Ambala et al. Absence of molecular evidence of filovirus circulating in bats and rodents in Laikipia North sub-County, Kenya: a cross sectional study. PAMJ-One Health. 2022;7:34. [doi: 10.11604/pamj-oh.2022.7.34.33231]
Available online at: https://www.one-health.panafrican-med-journal.com/content/article/7/34/full
Short communication 
Absence of molecular evidence of filovirus circulating in bats and rodents in Laikipia North sub-County, Kenya: a cross sectional study
Absence of molecular evidence of filovirus circulating in bats and rodents in Laikipia North sub-County, Kenya: a cross sectional study
![]() Peris Ambala1,2,&, Fatima Hussein2, Samson Mutura2,
Peris Ambala1,2,&, Fatima Hussein2, Samson Mutura2, ![]() George Gachara3, Nelson Menza3,
George Gachara3, Nelson Menza3, ![]() Lindsey McCrickard Shields4, Megan Vodzak4, Susan Murray4,
Lindsey McCrickard Shields4, Megan Vodzak4, Susan Murray4, ![]() Marc Valitutto4, Dawn Zimmerman4, Joseph Kamau2,4
Marc Valitutto4, Dawn Zimmerman4, Joseph Kamau2,4
&Corresponding author
In the recent decade, pathogenic zoonotic viruses have emerged in different geographical locations almost annually. These changes have led to new complex interactions between humans, animals and the environment, creating unique opportunities for pathogens unique opportunities to pass between hosts. Most emerging pathogens are RNA viruses such as filovirus. Numerous factors such as anthropogenic activities, changes in local ecosystem and climate change have contributed to this spillover. While Kenya has not reported any filovirus outbreaks in humans, a filovirus (i.e Bombali Ebola virus) have been detected in Kenyan bats, which have been implicated as reservoir hosts. The goal of this study was to detect and molecularly characterize known and novel filovirus circulating in bats and rodents in Laikipia North sub-County, Laikipia County, Kenya. In May 2018, a total of 477 samples (blood, oral and rectal swabs) were collected from 159 bats and 159 rodents in Laikipia North sub-County, Kenya. Ribonucleic acid was extracted from all samples and screened using consensus polymerase chain reaction targeting the long-gene of filovirus. All samples were negative. These results suggest that circulation of filovirus was uncommon during the month of May, 2018 in rodents and bats from Laikipia North sub-County. Considering our findings, future sampling should be conducted both longitudinally and with significantly larger sample sizes for a more in depth assessment of the prevalence of filoviruses in bats within the region studied.
Emerging and re-emerging viruses are of great concern not only on the African continent, but all over the world with a majority of the outbreaks occurring in the tropics including East Africa, Central Africa, West Africa [1]. These viruses cause outbreaks at unexpected times with severe consequences. In addition, there is a global movement of people across national borders which makes possible the transfer of infection to regions that were previously not known to harbor these viruses [2]. In this decade, examples of worldwide reported outbreaks of emerging and re-emerging viruses include severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), Ebola virus (EBOV), Middle East respiratory syndrome coronavirus (MERS-CoV) and influenza viruses. All of these viruses have emerged or re-emerged from animals [3]. Some of the most devastating viral disease outbreaks reported have been caused by viruses in the family Filoviridae [4]. Filovirus are amongst the highly pathogenic emerging and re-emerging viruses in humans, with a mortality rate of up to 90% in both humans and non-human primates [5]. So far, only three genera of this family (Marburg virus, Ebola virus and Cueva virus) have been described [4]. Members of this viral family are known to cause periodic outbreaks with severe hemorrhagic disease in both humans and non-human primates (NHPs) and respiratory disease in pigs [6]. The frequency of filovirus (Ebolavirus) outbreaks has increased over the last decade [7].
The first documented human filovirus outbreak was with Marburg virus in 1967, (Germany and Yugoslavia), from African green monkeys (Chlorocebus aethiops) imported from Uganda. Subsequently, in 1976 the first EBOV case in humans was reported in Nzara, South Sudan [8]. Thereafter, over 40 outbreaks of Ebolavirus and 10 outbreaks of Marburg virus have been reported in Africa and imported cases in the Netherlands, Yugoslavia, United States of America, Italy and United Kingdom [7,9]. To date, the most devastating EBOV outbreak occurred in West Africa in 2014-2016, with a case fatality rate as high as 62% [10]. In previous filovirus outbreaks, wildlife has been suspected to be the main source of filovirus emergence. The potential mammalian natural reservoir hosts of filoviruses include bats, duikers, rodents and NHPs [11]. Evidence shows that bats are the putative hosts and play a major role in transmission of filoviruses. Ebolavirus antibodies have been detected in several bats species in Africa (Guinea, Democratic Republic of Congo-DRC, Sierra Leone, Ghana, Liberia, South Africa, Uganda, Ivory Coast, Gabon), Asia (Philippines, Singapore, Bangladesh, India, China) and Europe (Spain) [8,12-14] and even isolated and sequenced from samples from Sierra Leone, China, DRC and Gabon [12,13,15]. Ebolavirus has also been serologically detected in several rodent species (Arvicanthis spp., Mastomys spp., Mastomys spp., Mus spp. and Praomys spp.) from the Central African Republic [16]. In fact, experimental studies have demonstrated the ability of rodents to adapt to filoviruses, meaning they can be infected [17]. Infected bats with filovirus pose as a risk of illness to humans.
Bats may represent a source of filovirus spillover in many African countries to both humans and other mammals. This may occur during hunting and preparation of bush meat or through direct exposure to fruits contaminated by infected saliva, feces or urine [18]. Kenya harbors a rich diversity of wildlife, lending it to becoming one of the top tourist destinations in Africa. Tourism accounts for a major percentage of Kenya´s gross development product (GDP), contributing approximately 9.7% of GDP in 2017 [19]. However, wildlife plays an important role in transmission of zoonotic diseases at the animal-human interface, which is extensive within Kenya. At the animal human interface in Laikipia North sub-County, it is not well understood what viral exchange occurs. Even though Kenya has not reported any filovirus outbreaks, filovirus antibodies have been detected in humans from the Central African Republic and DRC where no outbreak has been reported [20,21]. In addition, filoviruses have been detected serologically and have been isolated from bats and rodents, respectively. Moreover, in 2017, Kenya was listed as one of the countries at risk of imported filovirus (Ebolaviruses) despite the fact that it has only reported one outbreak of Marburg virus in 1980 [22]. We investigated the presence of known and novel filovirus RNA in bats and rodents from Laikipia North sub-County, Kenya.
Study design: this was a cross sectional study conducted in May, 2018 in Laikipia North sub-County, Laikipia, Kenya. The aim of this study was to detect the presence of known and novel filoviruses circulating in bats and rodents in Laikipia North County, Kenya.
Study area: Laikipia is a county in Kenya that lies between latitudes 0° 18" and 0° 51" North and longitude 36° 11" and 37° 24" East. Laikipia county is ranked as the second in wildlife population with a wide range of animals (both domestic and wild animals) that freely transverse the county [23,24]. Laikipia North has a human population size of 36,184 who interact with wild animals, thus increasing the risk of virus jump from animals to humans [25].
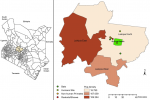
Sample collection: we employed convenience sampling technique, where we sampled the animals to saturation. To avoid bias, all the animals trapped were sampled. We sampled during the wet season, when insects and animals are found in large numbers. Insectivorous bats majorly feed on insects. In addition, during feeding, animals come in close contact, thus increasing the chances of filovirus infections. Blood clots, oral swabs and rectal swabs were collected from different species of bats and rodents from Ol Jogi and Ilmotiok villages from Laikipia North sub-County, Kenya (Figure 1). Depending on species of small mammals, different capture, bleeding techniques and sites were used. The animals were sampled in duplicate, collecting oral swabs, rectal swabs and blood. Rodents were captured using Sherman traps, while bats were captured using mist nets and harp nets. The animals were anesthetized using 0.4ml of isoflurane in cotton balls. Depending on the rodents' species blood was collected from the retro-orbital, lateral tail vein, jugular vein or ventral tail vein and in bats blood clots were collected from propetagial (cephalic) vein, the uropetagial (saphenous) vein, or the brachial vein [26,27]. The samples were stored in 500μl of virus transport media (VTM) while the other in 500μl of TRIzol™ media (LOT. No. 175806, Ambion® Life Technologies™). The samples were immediately placed in dry shippers containing liquid nitrogen, then shipped to the Institute of Primate Research Karen, where they were transferred to -80°C freezer.
RNA extraction and nested RT-PCR: RNA was extracted from each sample using DIRECT-Zol RNA-MiniPrep kit (Cat. #. R2052, Zymo Research). Following RNA extraction, the quality of RNA was confirmed using mitochondrial cytochrome-b RT-PCR [28]. Reverse transcription was performed using Superscript III first strand kit (Cat. # 18080-051, Invitrogen) for cDNA synthesis. Manufacturer´s instructions on use were followed. This was followed by a nested PCR using Platinum TM Taq DNA Polymerase kit (Cat. # 10966-026, Invitrogen). The amplification was carried out using PREDICT 11 filovirus protocol with modified primers from Zhai et al. 2007, from the conserved L-Gene of filovirus (Unpublished filovirus primers from University of California; Davis RT-PCR protocol [29]. The primers were modified to increase their sequence degeneracy in order to improve filovirus detection and discovery. The primary and secondary amplification conditions were 40 cycles, initial denaturation at 94°C for 5 minutes, denaturation at 94°C for 1 minute, annealing at 52°C for 1 minute, extension at 72°C for 1 minute, final extension at 7°C for 7 minutes. The positive control used in this study was a plasmid derived from reston filovirus (Filoreston, FiloR) provided by USAID PREDICT 11 project. For negative control we used PCR water that was used during the RT-PCR.
Ethical approval: this study was carried out in strict accordance with the recommendations in the Guide for Care and Use of Laboratory Animals in Kenya. Ethical approvals were granted by the Institute of Primate Research Ethical Review Committee, Ref. No: IERC/08/16. Research Permit was granted by National Council of Science and Technology (NACOSTI) permit number: NACOSTI/P/19/48092/2643.
Samples were collected during the wet season between 4th-11th May, 2018. The molecular prevalence of filovirus circulating in Laikipia North sub-County, was assessed in 477 samples from 159 bats and rodent species, each animal giving 3 samples (Table 1). A total of 100 bats (n=80 Chaerephon pumilus and n=20 Scotophilus dinganii) from the families Molossidae and Vespertilionidae were sampled. Each bat gave 3 samples i.e blood, oral swabs and rectal swabs. A total of 59 rodent species, 6 species from family Muridae and 1 species from family Nesomyidae, were sampled, each rodent giving 3 samples (blood, oral swabs and rectal swabs). All the blood samples tested negative for filovirus L-gene by RT-PCR. In addition, all the oral swabs and rectal swabs from the two animal taxa tested negative.
In the year 2017, Kenya was listed as one of the countries at risk of imported filovirus (Ebola viruses) despite the fact that it has never had outbreaks since 1980 (Marburg virus). Filovirus continue to emerge or re-emerge, with the most recent cases reported in April 2020 [30]. Filovirus ecology, reservoir hosts and spillover event to humans are still not completely understood. The suspected reservoir hosts (bats) are elusive and may be a source of infection to humans. Fruit bats have been linked through different index cases as potential sources of Marburg and Ebola viruses in Kenya and Uganda, respectively [31,32]. The viruses have been detected if not isolated from bats and experimentally in rodents. However, the role played by these animals as reservoir hosts and in natural ecology is not well understood. Nevertheless, filovirus infections in humans continue being a threat. To detect and identify animal species harboring filovirus in Laikipia North sub-County, Kenya, we tested 477 samples from 2 species of bats and 8 species of rodents. The aforementioned bats' species Chaerephon pumilus and bat family Vespertilionidae have tested positive for Bombali ebola virus by PCR (Sierra Leone) and serologically (Guinea, Cameroon and the DRC), respectively [15,33]. Although in our study all were negative, this is an important addition to the existing data on the epidemiology of filovirus in Kenya.
Filovirus ribonucleic acid (RNA) detection using molecular tools is rare. In a study by De Nys (2018) on bats, filovirus (Ebola viruses) viral RNA was not detected [33]. In experimental studies where Egyptian fruit bats (Rousettus aegyptiacus) were inoculated with Ebola virus strains (Sudan, Reston, Bundibugyo and Tai Forest) and Marburg virus, the bats did not shed EBOV while Marburg virus was shed through the oral and rectal route [34]. Similarly, in an experimental study, Paweska (2016) inoculated Egyptian fruit bats with EBOV but was not able to isolate the virus from oral or rectal swabs from any of the experimental animals, implying that the bats were not shedding the virus [35]. In nature, filovirus (Ebolavirus and Marburg virus) RNA has been detected by molecular tools in a couple of studies in fruit bats and insectivorous bats [12,36]. This is due to low viral load in the rectal and oral swabs [37]. Conducting concurrent serological assays would probably have indicated exposure of the animals to filoviruses.
In this study, samples were collected from Chaerephon pumilus (little free-tailed bat) and Scotophilus dinganii (Yellow African bat) which have been listed as a potential filovirus reservoir hosts [15]. In as much as we were not able to isolate filoviruses using molecular tools, we cannot rule out the possibility of these viruses circulating in bats from Laikipia North sub-County, Kenya because we only conducted molecular assays. Serological assays targeting antibodies would have demonstrated previous and current exposure of the animals to filoviruses in serum or plasma. In a review conducted by Olival (2014), many studies were not able to detect EBOV through PCR but were able to detect antibodies [38]. The negative molecular results in this study could have been due to low levels of detectable viral copies within the bats species, clearing of the virus by the bats naturally due to a robust immune response or the bats were never infected at all as argued by Lacroix 2021 [37].
Rodents have been suspected as being reservoir hosts to filovirus (EBOV) though not normally susceptible. In an experimental study by Morgan (1999), they demonstrated that EBOV can be isolated from experimentally infected rodent organs after several serial passages of the virus [39]. In addition, studies conducted by Pappalardo and co-workers, showed a few mutations at viral protein 24, VP24 (K142, L26F and L147P) and glycoprotein (S65 and D49) genes which are conserved in Ebolaviruses. These genes may contribute to the functionality of the virus and adaptation of the virus in rodents. This suggests that there is a probability of the viruses adapting to new rodent hosts, which may result in novel pathogenic human Ebolaviruses [40]. Our findings are similar to studies by Spengler (2015) where viral RNA from oral swabs from Guinea pigs model was not detected in ante-mortem animals [41]. In our study, we only sampled live animals.
Our findings show that animals sampled in Laikipia North sub-County in May 2018, did not harbor filoviruses. This may not be true due to the small sample size and a few bats and rodents' species we captured. A combination of capture techniques e.g. the use of both acoustic sampling, mist nets and turtle traps have been shown to capture large numbers of bats from different species. We believe that continued surveillance studies of filovirus should be conducted in small mammals longitudinally. The role of bats and rodents as reservoir hosts and in filovirus transmission still remains unclear, due to the fact that viral RNA detection is very rare. The identification of the reservoirs of filoviruses would aid in the development of strategies to prevent human outbreaks and reduce the impact of the viruses on animal species such as great apes, whose populations have been greatly threatened in endemic regions.
Funding: this study was supported by the US Agency for International Development Emerging Pandemic Threats PREDICT Project (cooperative agreement mo. GHN-A-OO-09-00010-00) and the National Research Fund (Ref. No: NRF/PhD/02/68) Kenya.
What is known about this topic
- Bats are reservoirs of filovirus;
- Ebolavirus antibodies and RNA has been detected in regions that have not reported filovirus outbreaks.
What this study adds
- The study describes the distribution of filovirus;
- The study suggests that rodents may not be the reservoir hosts of filoviruses;
- The study adds the different species of bats and rodents found in Laikipia County, Kenya.
The authors declare no competing interests.
All the authors have read and agreed to the final manuscript.
We thank the different teams that facilitated sample collection.
Table 1: animal species and types of samples collected
Figure 1: map of Kenya (sampling sites)
- Omoleke SA, Mohammed I, Saidu Y. Ebola viral disease in West Africa: a threat to global health, economy and political stability. J Public Health Afr. 2016 Aug 17;7(1):534. PubMed | Google Scholar
- Bogoch II, Creatore MI, Cetron MS, Brownstein JS, Pesik N, Miniota J et al. Assessment of the potential for international dissemination of Ebola virus via commercial air travel during the 2014 west African outbreak. The Lancet. 2015;385(9962):29-35. PubMed | Google Scholar
- Bonilla-Aldana DK, Jimenez-Diaz SD, Arango-Duque JS, Aguirre-Florez M, Balbin-Ramon GJ, Paniz-Mondolfi A et al. Bats in ecosystems and their wide spectrum of viral infectious potential threats: SARS-CoV-2 and other emerging viruses. Int J Infect Dis. 2021;102:87-96. PubMed | Google Scholar
- Jacob ST, Crozier I, Fischer WA, Hewlett A, Kraft CS, Vega MA de L et al. Ebola virus disease. Nat Rev Dis Primer. 2020;6(1):1-31. PubMed | Google Scholar
- Martina BE, Osterhaus AD. "Filoviruses": a real pandemic threat. EMBO Mol Med. 2009;1(1):10-18. PubMed | Google Scholar
- Kobinger GP, Leung A, Neufeld J, Richardson JS, Falzarano D, Smith G et al. Replication, pathogenicity, shedding, and transmission of Zaire ebolavirus in pigs. J Infect Dis. 2011;204(2):200-208. PubMed | Google Scholar
- WHO. Ebola virus disease. 2020. Accessed August 28, 2020.
- Languon S, Quaye O. Filovirus disease outbreaks: a chronological overview. Virol Res Treat. 2019;10:1178122X19849927. PubMed | Google Scholar
- WHO. Marburg virus disease. WHO. 2021. Accessed April 18, 2018.
- WHO. Origins of the 2014 Ebola epidemic. Accessed August 2, 2018.
- Peterson AT, Carroll DS, Mills JN, Johnson KM. Potential mammalian filovirus reservoirs. Emerg Infect Dis. 2004;10(12):2073-2081. PubMed | Google Scholar
- He B, Feng Y, Zhang H, Xu L, Yang W, Zhang Y et al. Filovirus RNA in fruit bats, China. Emerg Infect Dis. 2015;21(9):1675-1677. PubMed | Google Scholar
- Dovih P, Laing ED, Chen Y, Low DHW, Ansil BR, Yang X et al. Filovirus-reactive antibodies in humans and bats in Northeast India imply zoonotic spillover. PLoS Negl Trop Dis. 2019;13(10):e0007733. PubMed | Google Scholar
- Ogawa H, Miyamoto H, Nakayama E, Yoshida R, Nakamura I, Sawa H et al. Seroepidemiological prevalence of multiple species of filoviruses in fruit bats (Eidolon helvum) migrating in Africa. J Infect Dis. 2015 Oct 1;212 Suppl 2:S101-8. PubMed | Google Scholar
- Goldstein T, Anthony SJ, Gbakima A, Bird BH, Bangura J, Tremeau-Bravard A et al. The discovery of Bombali virus adds further support for bats as hosts of ebolaviruses. Nat Microbiol. 2018;3(10):1084. PubMed | Google Scholar
- Leroy E, Gonzalez JP, Pourrut X. Ebolavirus and other filoviruses. Wildl Emerg Zoonotic Dis Biol Circumst Consequences Cross-Species Transm. 2007;315:363-387. PubMed | Google Scholar
- Yamaoka S, Banadyga L, Bray M, Ebihara H. Small animal models for studying filovirus pathogenesis. Curr Top Microbiol Immunol. 2017;411:195-227. PubMed | Google Scholar
- Baudel H, Nys HD, Ngole EM, Peeters M, Desclaux A. Understanding Ebola virus and other zoonotic transmission risks through human-bat contacts: exploratory study on knowledge, attitudes and practices in Southern Cameroon. Zoonoses Public Health. 2019;66(3):288-295. PubMed | Google Scholar
- Rebecca MS. Tourism´s contribution to the economy of Kenya. Ann Tour Res. 1987;14(4):531-540. Google Scholar
- Mulangu S, Borchert M, Paweska J, Tshomba A, Afounde A, Kulidri A et al. High prevalence of IgG antibodies to Ebola virus in the Efé pygmy population in the Watsa region, Democratic Republic of the Congo. BMC Infect Dis. 2016;16(1):263. Google Scholar
- Gonzalez JP, Nakoune E, Slenczka W, Vidal P, Morvan JM. Ebola and Marburg virus antibody prevalence in selected populations of the Central African Republic. Microbes Infect. 2000;2(1):39-44. PubMed | Google Scholar
- Ministry of Health K. Press release on the Ebola viral disease preparedness measures in Kenya following the outbreak in Democratic Republic of Congo. 2021. Accessed September 27, 2021.
- Evans LA, Adams William M. Fencing elephants: the hidden politics of wildlife fencing in Laikipia, Kenya. Land Use Policy. 2016;51:215-228. Google Scholar
- Georgiadis NJ, Olwero JGN, Ojwang´ G, Romañach SS. Savanna herbivore dynamics in a livestock-dominated landscape: I, dependence on land use, rainfall, density, and time. Biol Conserv. 2007;137(3):461-472. Google Scholar
- Kenya National Bureau of Statistics. 2019 Kenya population and housing census volume II: distribution of population by administrative Units. 2019. Google Scholar
- CDC. Methods for trapping and sampling small mammals for virologic testing. 1995. Accessed September 27, 2021.
- Quesenberry K, Carpenter J. Ferrets, rabbits and rodents clinical medicine and surgery. 2nd Edition. 2004.
- Townzen JS, Brower AVZ, Judd DD. Identification of mosquito bloodmeals using mitochondrial cytochrome oxidase subunit I and cytochrome b gene sequences. Med Vet Entomol. 2008;22(4):386-393. PubMed | Google Scholar
- Zhai J, Palacios G, Towner JS, Jabado O, Kapoor V, Venter M et al. Rapid molecular strategy for filovirus detection and characterization. J Clin Microbiol. 2007;45(1):224-226. PubMed | Google Scholar
- WHO. Ebola virus disease-Democratic Republic of the Congo. 2021. Accessed October 13, 2021.
- Towner JS, Amman BR, Sealy TK, Carroll SAR, Comer JA, Kemp A et al. Isolation of genetically diverse marburg viruses from Egyptian fruit bats. PLOS Pathog. 2009;5(7):e1000536. PubMed | Google Scholar
- Kuzmin IV, Niezgoda M, Franka R, Agwanda B, Markotter W, Breiman RF et al. Marburg virus in fruit bat, Kenya. Emerg Infect Dis. 2010;16(2):352-354. PubMed | Google Scholar
- De Nys HM, Kingebeni PM, Keita AK, Butel C, Thaurignac G, Villabona-Arenas CJ et al. Survey of Ebola viruses in frugivorous and insectivorous bats in Guinea, Cameroon, and the Democratic Republic of the Congo, 2015-2017. Emerg Infect Dis. 2018;24(12):2228-2240. PubMed | Google Scholar
- Jones MEB, Schuh AJ, Amman BR, Sealy TK, Zaki SR, Nichol ST et al. Experimental inoculation of Egyptian Rousette bats (Rousettus aegyptiacus) with viruses of the ebolavirus and Marburgvirus Genera. Viruses. 2015;7(7):3420-3442. PubMed | Google Scholar
- Paweska JT, Storm N, Grobbelaar AA, Markotter W, Kemp A, Jansen van Vuren P. Experimental Inoculation of Egyptian Fruit Bats (Rousettus aegyptiacus) with Ebola Virus. Viruses. 2016 Jan 22;8(2):29. PubMed | Google Scholar
- Forbes KM, Webala PW, J��skel�inen AJ, Abdurahman S, Ogola J, Masika MM et al. Bombali virus in mops condylurus Bat, Kenya. Emerg Infect Dis. 2019 May;25(5):955-95 PubMed
- Lacroix A, Mbala Kingebeni P, Ndimbo Kumugo SP, Lempu G, Butel C, Serrano L et al. Investigating the circulation of Ebola viruses in bats during the Ebola virus disease outbreaks in the Equateur and North Kivu Provinces of the Democratic Republic of Congo from 2018. Pathogens. 2021;10(5):557. PubMed | Google Scholar
- Olival KJ, Hayman DTS. Filoviruses in bats: current knowledge and future directions. Viruses. 2014;6(4):1759-1788. PubMed | Google Scholar
- Morvan JM, Deubel V, Gounon P, Nakoune E, Barriere P, Murri S et al. Identification of Ebola virus sequences present as RNA or DNA in organs of terrestrial small mammals of the Central African Republic. Microbe Infect. 1999;1(14):1193-201. PubMed | Google Scholar
- Pappalardo M, Reddin IG, Cantoni D, Rossman JS, Michaelis M, Wass MN. Changes associated with Ebola virus adaptation to novel species. Bioinformatics. 2017;33(13):1911-1915. PubMed | Google Scholar
- Splengler JR, Chakrabarti AK, Coleman-McCray JD, Martin BE, Nichol ST, Spiropoulou CF et al. Utility of oral swab sampling for Ebola virus detection in Guinea Pig Model. Emerg Infect Dis. 2015;21(10):1816-1819. PubMed | Google Scholar