Mortality rate and its determinants among colorectal cancer patients in comprehensive specialized hospitals, Ethiopia: a retrospective cohort study
Bantalem Tilaye Atinafu, Worku Misganaw Kebede, Tefera Mulugeta Demlew, Yared Asmare Aynalem, Wondimeneh Shibabaw Shiferaw, Fetene Nigussie Tarekegn, Getaneh Baye Mulu
Corresponding author: Worku Misganaw Kebede, Department of Nursing, Health Science College, Debre Berhan University, Debre, Ethiopia 
Received: 20 Apr 2021 - Accepted: 29 Mar 2022 - Published: 31 Mar 2022
Domain: Chronic disease prevention
Keywords: Incidence, mortality, colorectal cancer, determinants, Tikur Anbessa specialized hospital, Ethiopia
©Bantalem Tilaye Atinafu et al. PAMJ-One Health (ISSN: 2707-2800). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Bantalem Tilaye Atinafu et al. Mortality rate and its determinants among colorectal cancer patients in comprehensive specialized hospitals, Ethiopia: a retrospective cohort study. PAMJ-One Health. 2022;7:26. [doi: 10.11604/pamj-oh.2022.7.26.29412]
Available online at: https://www.one-health.panafrican-med-journal.com/content/article/7/26/full
Research 
Mortality rate and its determinants among colorectal cancer patients in comprehensive specialized hospitals, Ethiopia: a retrospective cohort study
Mortality rate and its determinants among colorectal cancer patients in comprehensive specialized hospitals, Ethiopia: a retrospective cohort study
Bantalem Tilaye Atinafu1, Worku Misganaw Kebede1,&, Tefera Mulugeta Demlew2, ![]() Yared Asmare Aynalem1,
Yared Asmare Aynalem1, ![]() Wondimeneh Shibabaw Shiferaw1,
Wondimeneh Shibabaw Shiferaw1, ![]() Fetene Nigussie Tarekegn1, Getaneh Baye Mulu1
Fetene Nigussie Tarekegn1, Getaneh Baye Mulu1
&Corresponding author
Introduction: the mortality rate of colorectal cancer rises rapidly in low- and middle-income countries. Thus, this study aimed to assess the incidence of mortality and its determinant factors among colorectal cancer patients in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia.
Methods: a retrospective cohort study was done among confirmed diagnoses of patients registered in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia, between January 1st, 2013 to January 31st, 2016, followed up to January 31st, 2018. A total of 434 study participants were selected using census sampling. We performed training and pretest before the data was collected from patient record reviews. Data was entered using epi-data 4.2 and analyzed by STATA 14. Basic descriptive statistics were done. Kaplan Meier failure curve was used to show mortality differences among covariates. Bivariate and multivariable cox proportional hazard regression was done to show the net effect of independent variables.
Results: among 434 study subjects, the cumulative incidence of death of colorectal cancer patients over the six-year follow-up period was 151 (34.8%). The overall mortality rate for diagnosed colorectal cancer patients during 818 person-year observations was 18.3 per 100 (95% CI:15.7-21.7) person-year follow-up. Age ≥60 years old, marital status, comorbidity, stage, and grade of tumor were found to be significant predictors of mortality among confirmed diagnoses of colorectal cancer.
Conclusion: the incidence of mortality among colorectal cancer patients was high. It is essential to establish screening protocol and provide brief interventions, where appropriate.
Globally, colorectal cancer (CRC) is a major public problem, which had a wide variation overtime in different geographical areas [1]. The significantly increased incidence has been seen in developing countries in contrast to developing countries due to sedentary behaviors and lifestyles [2]. Evidence showed that CRC was estimated to be the second cause of cancer-related death due to an aging population and population growth, which contributes 17%, 12%, and causes of colorectal cancer incidence and mortality, respectively [3]. According to GLOBOCAN 2018 estimation, the incidence of colorectal cancer was 1.80 million of those 881 000 mortality cases were estimated. It is the deadliest disease that varies depending on the economic development of nations. The age-standardized mortality was about 12.8/100,000 and 5.7/100,000 among males in high and low human development index nations, respectively. By the year 2030, colorectal cancer is predicted to rise by 60%, to over 2.2 million new cases and 1.1 million annual deaths as a result of increased population, lifestyle factors, economic status [4,5].
In sub-Saharan Africa (the crude incidence of colorectal cancer was 4.04 per 100 000 population for both men and women; around 24,711 new cases were estimated yearly [6]. A study done in Ghana revealed that all the patients diagnosed at stage IV were not survived during five years follow-up [7]. The incidence and mortality rates are still rising rapidly in many low-income and middle-income countries, which are linked to ongoing societal and economic development [4,8]. A personal lifestyle, clinical status, and hereditary mutation where risk factors that determine the incidence of mortality [9]. In Ethiopia, the Federal Ministry of health emphasizes non-communicable diseases such as cancer to reduce the incidence and mortality. However, little is known about the incidence of mortality and associated factors. That means interventions to enhance survival and reduce mortality in colorectal cancer lack the necessary empirical pieces of evidence. As a result, there could be an evidence-based decision-making gap about colorectal cancer, such as prioritizing interventions, estimating the survival rate of patients, and supporting the planning systems of cancer control and prevention program. Hence, this study aimed to assess the incidence of mortality and its predictors among colorectal cancer patients in a comprehensive specialized hospital in Ethiopia.
Study area and period study design: a follow-up study among colorectal cancer patients was conducted in Tikur Anbessa Specialized Hospital oncology unit. Addis Ababa is Ethiopia´s capital city with ten sub-cities and 116 districts. Tikur Anbessa Specialized Referral Hospital is the most significant referral and teaching public hospital in Ethiopia, established in 1972. It is the training center of health professionals, including undergraduate and postgraduate medical students, dentists, nurses, pharmacists, laboratory technicians, and other paramedics. Tikur Anbessa Specialized Hospital(TASH), is a tertiary teaching hospital with 800 beds and gives about 370,000 to 400,000 patients per year. The oncology center at the hospital is the only referral center. The oncology unit is giving more than 60,000 cancer patients annually and has an outpatient, in-patient (33 beds), radiotherapy, and chemotherapy and surgery care service. There are six senior oncologists, 25 residents and 30 nurses and six oncology nurses and eight pharmacists, five radiologists, four medical physicists working in the unit. The study was conducted from February to March 2019 at the oncology unit of TASH, College of Health Sciences, and Addis Ababa University, Ethiopia.
Population: the source population consisted of all medical records of colorectal cancer patients in the TASH Oncology Unit. The study population includes all medical records of colorectal cancer patients in TASH who were diagnosed from January 1st, 2013 to December 30th, 2016 who fulfill eligibility criteria. We had all medical records of confirmed colorectal cancer patients at TASH during the (2013-2016) period. These included incomplete patient charts, missed patients' charts during the data collection period, and referred patients with confirmed diagnosis TASH for advanced management were excluded.
Sample size determination, sampling procedure, and study variables: in the beginning, we identified all medical records of a confirmed diagnosis of colorectal cancer patients registered from January 1st, 2013 to December 30th, 2016. From 700 identified medical records of colorectal cancer patients, 191 incomplete charts, missed charts 72 at data collection time, and three were referred for advanced treatment (radiation) were excluded from the study. Finally, all study participants who fulfill the inclusion and exclusion criteria from January 1st, 2013 to December 30th, 2016, were selected. The dependent variable was the incidence of death. Age, sex, family history, marital status, residence, insurance status, smoking status, alcohol consumption, BMI and comorbidity, grade at diagnosis, stage at diagnosis, a primary site, and histologic type and treatment were variables of interest that were extracted from the review record.
Data collection methods and procedures: we used a data abstraction format adapted from different literature and Addis Ababa city cancer register to collect data and necessary information from patients' medical files and the records section in the cancer center of the TASH. Senior experts to the area of study for content validity examined the checklist. Data abstraction is designed based on study objectives and contains socio-demographic characteristics and clinical and treatment characteristics. The clinical stage at diagnosis was assigned to each patient by using America joint committee of cancer (AJCC) (Stage 0: carcinoma in situ, no lymph node, and no metastasis, Stage I: tumor invades muscularis propria, submucosa, no lymph node, and no metastasis, Stage II: tumor invades muscularis propria, penetrates to the surface of the visceral peritoneum, adherent to other organs or structure, no lymph node, and no metastasis, Stage III: tumor metastasis in seven or more regional lymph nodes, Stage IV: tumor metastasis into different organs. The Nottingham Grading System, grade assessed histological grade of CRC (1-well differentiated/2 moderately differentiated/3-poorly differentiated/undifferentiated). We reviewed all charts of CRC patients, diagnosed between January 1st, 2013 to December 31st, 2016 at TASH from cancer registries. We selected the records of all study participants according to the eligibility criteria. Before collecting the data, we reviewed we identified documents (both baseline and follow-up records), death certificates complemented by registration from their medical record number. These data collectors were working at the cancer treatment center, extracted and reviewed the charts.
Data quality control: we assured data quality by designing proper data abstraction tools. Experienced researchers evaluated the adapted checklist. A pretest was employed on 5% of the sample size with a structured checklist two weeks before the actual study to check usually recorded variables on the patient´s medical record. Consequently, we reduced unrecorded variables from the checklist, and others arranged as per usual records of those variables. The data collector and supervisor have experienced MSc oncology nurses working in this unit, and training was given for them effectively. We gave one-day training concerning the data abstraction tool and data collection process for both data collectors and supervisors. During the data collection time, close supervision and monitoring were carried out by supervisors and investigators to ensure the quality of the data. We attended daily evaluation of the data for completeness and encountered difficulties on the time of data collection accordingly. Double date entry was done using Epi-data version 4.2 software. Finally, the supervisor and investigator checked all the collected data for its completeness and consistency during the data management, storage, and analysis. Consistency was examined through a random selection of cards by the principal investigator.
Data processing and analysis: the collected data were coded, entered, edited, and cleaned using EPI-data 3.1 and exported to STATA 14.0 statistical software for analysis. Frequencies, proportions, and descriptive statistics were used to describe the study population related to relevant variables. Kaplan Meier analysis was used to identify the overall hazard rates and median hazard time. Differences in survival among different variables were compared using the log-rank test. We calculated the incidence rate of death for the entire study period. Subsequently, the number of mortalities within the follow-up was divided by the total person-time at risk on follow-up and reported per 100 person-year. Before running the Cox regression model, the assumption of proportional-hazard was performed. The cox-proportional hazard model assumption was checked using the Schoenfeld residual test, and we considered variables with P-value >0.05.to fulfill the assumption. We included variables with a significance level below 0.2 in the bivariate Cox regression model to determine the association between the dependent and explanatory variables. Tables and figures exemplified the result of the study.
Individual related factors: between January 1st 2013, to December 31st, 2016, 700 colorectal cancer patients were enrolled at Tikur Anbessa Specialized Hospital, from which 434 were eligible for this study. About 42.6% were females, and 41.2% of patients were age below 40 years old. Nearly one-four of patients' BMI was below 18.5. Regarding on the distribution of patient 42.6% from Addis Ababa, 26.5% from Oromiya, 12.4% from Amhara, 9.0% from SNNP and 6.2% from Tigray. Around 24.4% of patients had comorbid conditions; of those, 6.9% had hypertension, 3.7% had diabetes mellitus, and 3.5% had retroviral infections (Table 1).
Clinical and treatment-related characteristics: the majority (79.3%) of cases were adenocarcinoma types of tumor. Nearly half of the patients were diagnosed as well-differentiated, followed by a moderately differentiated tumor. About 62.7% of patients were diagnosed at a late stage of cancer, whereas 37.3 were diagnosed at the early stage of the tumor. Regarding treatment given, around 27.6% of patients received chemotherapy as an adjuvant to surgery because patients were diagnosed with the late presentation of the tumor (Table 2).
The mortality rate among colorectal cancer patients: a total of 621 colorectal cancer patients were followed for six years. The overall mortality rate for diagnosed colorectal cancer patients registered at TASH during 818 person-year observations was 18.5 per 100 (95% CI:15.7-21.7) person-year follow-up. The cumulative incidence of death of colorectal cancer patients over the six-year follow-up period was 151(34.8%), while 283 (65.2%) were either lost from follow-up or alive up to the end of the study.
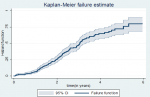
The overall failure rate of colorectal cancer patients: as the Kaplan Meier failure estimate curve presented, the overall hazard rate was 80.05% at 6 years follow-up. The estimated cumulative hazard rates of colorectal cancer patients at 1, 2, 3, 4, and 5 years were 6.7%, 25.7%, 48.2%, 65%, and 76.06%, respectively. The probability of hazard is the lowest on the first day of diagnosis of colorectal cancer, but it is relatively raised later as follow-up time increases. In this study, we found the highest mortality rate between 3 years and 3 months and five years of a confirmed diagnosis of colorectal cancer (Figure 1).
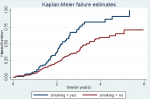
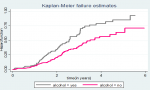
Hazard function and comparison of hazard functions for different categorical variables: the Kaplan-Meier failure estimator curve estimates hazard function among different groups of covariates to make comparisons. We constructed separate graphs of the estimates of the Kaplan-Meier hazard functions. The pattern that one hazard function lying above another group demarcated by the upper curve has higher mortality than the group demarcated by the lower curve, which had a more favorable survival capability than the group demarcated by the lower curve. This difference was explained statically by the log-rank test. Hence, the presence of any significant difference in hazard time was considered in this study. The test statistics obtained from the log-rank test clearly showed a significant difference in the hazard curve for different categorical variables. The study found that the hazard rate of colorectal cancer was higher among males (84.4%; CI: 67.9- 95.2) than the hazard rate of females (73.3%; CI: 53.6 - 89.6). Those colorectal cancer patients with comorbid conditions had higher hazard rates than those without comorbid conditions. Patients who smokers cigarettes had a higher mortality rate than nonsmokers; similarly, alcohol consumer also had higher hazard rate than non-alcohol consumers (Figure 2,Figure 3). The 3- and 5-years mortality rate for patients diagnosed as stage I colorectal cancer was 8.5% and 15.1%; For stage II were 2.8%, 36.8%, 52.8%: for stage III were 7.9%, 51.8%, 100%: for stage IV were 11.4%, 74.1%, 100% respectively. Those diagnosed as differentiated tumors had lower mortality rates (75.2%; CI: 51.4-91.6) than undifferentiated types (89.5%; CI: 76.9-96.9). Regarding the histology nature of the tumor at the time of diagnosis, the patient diagnosed as adenocarcinoma had a hazard rate of 76.1%. In contrast, mucinous adenocarcinoma and signet ring cell carcinoma have not existed in 6 years. This was indicated as a lower hazard rate among patients with adenocarcinoma. A patient whose age < 40 had 6.2%,41.6%, 76.3% at 1,3, and 5 years of follow-up was higher than the age group above 40 years old.
Predictors of colorectal cancer mortality: the relationship between the explanatory variables and incidence of mortality was analyzed using Cox proportional hazard regression model. We fitted variables such as sex, age (60-69 and > 70 years), marital status, smoking, alcohol consumption, comorbidity, stage, grade, and histology type at p-value < 0.25 to prevent the negative confounding effect. Hence, in the multivariable cox proportional hazards model; age group ≥60 years, comorbidity, marital status, stage, and grade were significant predictors of colorectal cancer mortality (P-value<0.05). Multivariable analysis showed that Patients whose marital status was widowed 1.7 times (AHR=1.7, CI: 0.81-3.7) and divorced 2.7 times (AHR=2.7, CI: 1.6-4.5) were more likely to die than single marital status. Age group ≥ 70 years were 2.2 times more likely to die (AHR: 2.2, CI: 1.2-4.1) than age below 40 years old as a reference. Those colorectal cancer patients who had comorbidity were 1.9 times more likely to die than non-comorbid patients (AHR: 1.9, CI: 1.3-2.7). Patients who were diagnosed at clinical stage III were 9.5 times more likely to die than those who were diagnosed at clinical stage I (AHR: 9.5, CI: 2.8-31.8) (Table 3).
Ethics approval and informed consent: ethical clearance for the proposed study was obtained from Addis Ababa University, School of Nursing, and Midwifery research ethical committee with Institutional Review Board(IRB) number of 017/19/SNM. A letter of cooperation was written to the Black Lion Specialized Hospital and concerned bodies. Then we obtained signed written informed consent from the medical director and cancer treatment center focal person of Black Lion Specialized Hospital. Confidentiality of the information was kept throughout the study by excluding names and patient record numbers as identification in the data extraction form, and the data were used only for the proposed research. To maintain confidentiality; all collected data were coded and locked in a separate room before entering the computer. After entering the computer, the data were locked by password, and the data were not disclosed to any person other than the principal investigator. The study has followed the Helsinki declaration of ethical principles.
This study aimed to assess the incidence of mortality and its determinant factors among colorectal cancer patients in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia. We found age group ≥60 years, comorbidity, marital status, stage, and grade to be significant predictors of colorectal cancer mortality incidence. More than half (62.7%) of the patients are diagnosed at a late stage, which is similar to the 2015 national cancer control program report in Ethiopia (80%) [10]. Perhaps due to low awareness of signs and symptoms, inadequate screening and early detection and treatment services, insufficient diagnostic facilities, and poorly structured referral. This study revealed that the cumulative incidence was 34.8% and a mortality rate of 18.5 per 100 person-years. This finding is lower than a study conducted in Iran [11] and China [12], but higher than the study conducted in sub-Saharan countries and the United States cancer report [6,13,14]. The difference could be due to follow-up time variation, absence of early screening and detection-besides, alcohol, tobacco smoking, and physical inactivity attribute. The incidence of overall mortality of colorectal cancer patients was 80.05%; this finding is higher than the study conducted in Canada (61%) [15] and in Yazd (60.2%) [16]. This difference might be improved screening and diagnostic modalities and advanced treatment intervention, including the multidisciplinary involvement team during service delivery.
In this retrospective cohort study, the overall median survival time of confirmed diagnosis of colorectal cancer patients was 34.8 months. This result is in line with the study conducted in South Iran (36.06 months) [17]. In contrast, it is lower than other studies which were conducted in Malaysia (42 months) [9], North Iran (40.5 months) [18], and Greece (98 months) [19]. However, median survival was higher than a study conducted in Ghana was 15 months [7]. The lower median survival time compared with the study in Ghana may be due to differences in a follow-up study, early detection, health-seeking behavior, and treatment adherence. This study showed that the 1, 3, and 5 years mortality of colorectal cancer patients were 6.7%, 48.2% and 76.06% respectively. This finding is consistent with other previous studies which have been conducted in Iran (93.9%, 50.3%, and 27.2%) at 1, 3 and 5 years. However, this finding was lower than Taiwan (95.3%, 79.4, 68.7), Kurdistan (87%, 69%,57%,42%, 33%), Iran (71%,52%.44%) Malaysia (73.8-76%,52.1-51.7%, 40.4-45.4%) at 1, 3.5 years and Newzland (51%), Jordan (58.2), Saudi Arabiya (44.6%) at 5 years. In addition, this finding is higher the study conducted in Ghana (64%,40%, 21%, 16%, 16%) [7,9,17,18,20-25].
This discrepancy may be due to a lack of early screening programs, a higher proportion of advanced-stage cancer at the time of diagnosis, lack of specialized care, and delay in receiving care. Other possible explanations may be that health facilities exist at the city centers, but patients from the countryside can't access them. As a result of economic constraints and being far from the health facility, patients may delay in diagnosis and treatment. Also, the discrepancy might be due to differences in methodology, the study period, individual differences, and differences in clinical presentation. This study revealed that being diagnosed at Stage IV was a strong predictor of mortality, similar to a study conducted in Canada [15]. Similarly, previous studies conducted in Iran were clinically diagnosed stage [26]. A study in Iran revealed that stage was a significant factor for colorectal cancer mortality for patients diagnosed as early-stage at baseline were less likely to die than late-stage at baseline [11]. The possible explanation might be a delay in a timely recipient of treatment, the presence of comorbidities. The current study showed that colorectal cancer patients diagnosed as undifferentiated tumor grade were 1.9 times at high hazard to die than those who were the well-differentiated type of tumor due to the aggressive nature of the undifferentiated type of tumor tend to have poor survival outcomes. The evidence showed that the undifferentiated tumor tends to grow and spread more quickly than a well-differentiated and moderately differentiated tumor. Besides, the undifferentiated tumor is associated with a high-grade tumor [27]. This study revealed that we also found marital status to be a significant factor in colorectal cancer mortality. This is in line with the previous research done in China [28-30]. Socio-demographic factors such as marital status were a well-known determinant factor for disease progression associated with social support. The married people had better social support, which made it more beneficial to the psychological dimension, early detection, and screening, and had better adherence to treatment intervention. However, the current study showed married people were more likely to die than single marital status. This difference might be due to the greater number of married patients and those married at an elder age.
This study showed that we found older age to be statistically significant to colorectal cancer mortality incidence among younger age counterparts. This is due to the refusal state of treatment intervention at an older age, the presence of comorbidity. Regarding comorbidity, it was found to be statically significant for mortality incidence, which is similar to a study conducted in Australia [31], and Nigeria, [32]. Cancer patients with comorbidity cause rapid tumor growth, which tends to diagnose lately, drug interaction, toxicities, reduce the effectiveness of treatment interventions, or reduce compliance [33]. The presence of the coexisting medical condition and the primary disease affects the treatment option and patient survival outcome. Thus, those patients who had comorbid conditions are less likely to receive standard care than non-comorbid patients because of the toxicity and side effects of treatment and the treatment approaches. Patients with comorbidity conditions are less likely to be treated by curative procedures as a result; comorbidity makes patients prone to poor survival outcomes. Moreover, the comorbid condition increases the disease´s progression, increases the recurrence of the tumor, and severely affects the immune status of the patient [34]. The presence of comorbidity also poses on care delivery. The health care needs of these patients require diverse experts, significant care coordination to optimize the treatment. This is also associated with an increased burden on health care costs [35].
In the current study alcohol consumption was found to be a significant factor for mortality among colorectal cancer patients. This finding was similar to the previous study conducted in Scotland [36]. As far as alcohol intake as a risk for cancer, the intake of alcohol complicates the progression of the tumor. Review evidence from various clinical and control trials revealed that alcohol has a diverse effect on body systems. For example, alcohol consumption associated with cardiomyopathy, cardiac hypertrophy, atherosclerosis, hormonal dysregulation, gastrointestinal lining irritation, etc. enables the tumor to progress rapidly [37]. As a result of oxidative and non-oxidative metabolism, Alcohol leads to disruption of hemostasis; the release of endotoxin, which induces inflammation, leads to local and systemic effects resulting in mucosal immunity suppression [38]. Moreover, alcohol consumption also causes cellular damage [39]. This study found smoking status a significant factor for mortality among colorectal cancer. This finding was similar to the previous study conducted in Norway [40], and the District of Columbia, and Puerto Rico [41], and Germany [42]. Evidence bared that smoking causes alteration of mucosal proliferation, increases the viral and bacterial envision, and alters the immune system of GI mucosa [43]. The nicotinic substance found in smoking causes deteriorates the harmful effects of aggressive factors and diminishes the protective effect of protective factors. This leads to the rapid progression of the tumor [44]. Moreover, those who smoke cigarettes and consume Alcohol had a high probability of other underlying medical problems. As a result, this pathological mechanism worsens patients´ survival outcomes [40]. The strength of the follow-up study was reasonably long, which makes the findings representative. Data were collected by oncology nurses who had an essential role in the data quality. Limitations include selection bias possibly introduced during secondary data collection because patients with incomplete records were excluded. Cause-specific mortality was not determined, as data on the specific cause of death did not register. Biological biomarkers, treatment adherence, physical exercise, the cycle of chemo, aim of treatment, educational status, and multidisciplinary care variables that might have a significant association with mortality could not be found on the medical cards and were not assessed.
The overall incidence of mortality among colorectal cancer patients was 80.05% at 6 years of follow-up. The findings revealed a higher mortality rate among confirmed diagnosis of colorectal cancer patients in Tikur Anbessa Specialized Hospital as compared with those of high- and middle-income countries. Age ≥60 years old, marital status, comorbidity, stage, and grade of tumor found significant predictors of mortality among confirmed diagnoses of colorectal cancer. This study recommends an early colorectal cancer screening and detection program, specializing. Further research could be conducted by including laboratory findings, societal and health system-related factors, and molecular biomarkers.
What is known about this topic
- Colorectal cancer is the third common cancer and the second cause of mortality among males and females globally;
- Mortality rates are still rising rapidly in many low and middle-income countries;
- First most common cancer among the male population in Ethiopia.
What this study adds
- No current data on the mortality rate of colorectal cancer;
- To guide the national cancer control program, to support the planning systems for better cancer control and prevention program;
- To implement early detection, prioritize intervention, and make an evidence-based decision.
The authors declare no competing interests.
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work. All the authors have read and agreed to the final manuscript.
Our gratitude goes to the School of Nursing and Midwifery, College of Health Sciences, Addis Ababa University, for giving us the chance to do this research. Authors� thanks also go to Black Lion Specialized Hospital Manager, all Oncology Unit staff, card room officers, and data collectors for their cooperation during data collection.
Table 1: socio-demographic characteristics of colorectal cancer patients in TASH oncology department, Addis Ababa, Ethiopia, 2019 (n=434)
Table 2: clinicopathological and treatment-related characteristics of colorectal cancer patients in TASH oncology department, Addis Ababa, Ethiopia, 2019 (n=434)
Table 3: bivariate and multivariable cox regression analysis of colorectal cancer patients in Tikur Anbessa specialized hospital, Addis Ababa, Ethiopia, 2019 (n=434)
Figure 1: the cumulative Failure rate of colorectal cancer patients diagnosed at Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia, 2019
Figure 2: the Kaplan-Meier failure function compare failure time of colorectal cancer patients with different categories of baseline smoking in Tikur Anbessa specialized hospital, Addis Ababa, Ethiopia, 2019
Figure 3: the Kaplan-Meier failure function compares hazard time of colorectal cancer patients with different categories of baseline alcohol consumption in Tikur Anbessa specialized hospital, Addis Ababa, Ethiopia, 2019
- Kanyike AM, Olum R, Kajjimu J, Ojilong D, Akech GM, Nassozi DR et al. Acceptance of the coronavirus disease-2019 vaccine among medical students in Uganda. Tropical medicine and health.2021;49(1):37-37. PubMed | Google Scholar
- Favoriti P, Carbone G, Greco M, Pirozzi F, Pirozzi RE, Corcione F. Worldwide burden of colorectal cancer: a review. Updates in Surgery. 2016 Mar;68(1):7-11. PubMed | Google Scholar
- Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Abate D, Abbasi N, Abbastabar H, Abd-Allah F et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2016: a systematic analysis for the global burden of disease study. JAMA Oncology. 2018;4(11):1553-68. PubMed | Google Scholar
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians. 2018;68(6):394-424. PubMed | Google Scholar
- Siegel RL, Torre LA, Soerjomataram I, Hayes RB, Bray F, Weber TK et al. Global patterns and trends in colorectal cancer incidence in young adults. Gut. 2019;68(12):2179-85. PubMed | Google Scholar
- Graham A, Davies Adeloye LG, Theodoratou E, Campbell H. Estimating the incidence of colorectal cancer in sub-Saharan Africa: a systematic analysis. Journal of global health. 2012 Dec;2(2):020404. PubMed | Google Scholar
- Agyemang-Yeboah F, Yorke J, Obirikorang C, Nsenbah Batu E, Acheampong E, Amankwaa Frimpong E et al. Colorectal cancer survival rates in Ghana: a retrospective Hospital-Based Study. PloS one. 2018;13(12):e0209307. PubMed | Google Scholar
- Magaji BA, Moy FM, Roslani AC, Law CW. Survival rates and predictors of survival among colorectal cancer patients in a Malaysian Tertiary Hospital. BMC Cancer. 2017;17(1):339. PubMed | Google Scholar
- Rawla P, Sunkara T, Barsouk A. Epidemiology of colorectal cancer: Incidence, mortality, survival, and risk factors. Prz Gastroenterol. 2019;14(2):89-10. PubMed | Google Scholar
- Health FMo. Disease Prevention And Control Directorate. Ethiopia National Cancer Control Plan. 2016-2020 2015:14.
- Ahmadi A, Mosavi-Jarrahi A, Pourhoseingholi MA. Mortality determinants in colorectal cancer patients at different grades: a prospective, cohort study in Iran. Asian Pacific journal of cancer prevention: APJCP. 2015;16(3):1069-72. PubMed | Google Scholar
- Gu MJ, Huang QC, Bao CZ, Li YJ, Li XQ, Ye D et al. Attributable causes of colorectal cancer in China. BMC cancer. 2018;18(1):38. PubMed | Google Scholar
- Ansa BE, Coughlin SS, Alema-Mensah E, Smith SA. Evaluation of colorectal cancer incidence trends in the United States (2000-2014). Journal of clinical medicine. 2018;7(2):22. PubMed | Google Scholar
- ACS. Cancer Facts & Figures 2018. Atlanta, Ga: American Cancer Society. Atlanta. 2018.
- Yu Y, Carey M, Pollett W, Green J, Dicks E, Parfrey P ,et al. The long-term survival characteristics of a cohort of colorectal cancer patients and baseline variables associated with survival outcomes with or without time-varying effects. BMC medicine. 2019;17(1):150. PubMed | Google Scholar
- Vakili M, Aghakoochak A, Pirdehghan A, Shiryazdy M, Saffarmoghadam A. The survival rate of patients with colorectal cancer in Yazd during 2001-2011. SSU_Journals. 2014;22(3):1187-95. PubMed | Google Scholar
- Mehrabani D, Almasi-Hashiani A, Moshfeghi K, Khedmati E. Survival rate and its predictors in colorectal cancer patients, Southern Iran. Middle East J Sci Res. 2012;12(8):1072-7. Google Scholar
- Semnani S, Noorafkan Z, Aryaie M, Sedaghat SM, Moghaddami A, Kazemnejhad V et al. Determinants of healthcare utilisation and predictors of outcome in colorectal cancer patients from Northern Iran. European Journal of Cancer Care. 2016;25(2):318-23. PubMed | Google Scholar
- Zacharakis M, Xynos ID, Lazaris A, Smaro T, Kosmas C, Dokou A et al. Predictors of survival in stage IV metastatic colorectal cancer. Anticancer Research. 2010;30(2):653-60. PubMed | Google Scholar
- Alsanea N, Abduljabbar AS, Alhomoud S, Ashari LH, Hibbert D, Bazarbashi S. Colorectal cancer in Saudi Arabia: incidence, survival, demographics and implications for national policies. Annals of Saudi Medicine. 2015;35(3):196-202. PubMed | Google Scholar
- Hoseini S, Moaddabshoar L, Hemati S, Mohammadianpanah M. An overview of clinical and pathological characteristics and survival rate of colorectal cancer in Iran. Ann Colorectal Res. 2014;2(1):e17264. Google Scholar
- Sharkas GF, Arqoub KH, Khader YS, Tarawneh MR, Nimri OF, Al-Zaghal MJ et al. Colorectal cancer in Jordan: Survival rate and its related factors. Journal of Oncology. 2017;2017:3180762. PubMed | Google Scholar
- Sharples KJ, Firth MJ, Hinder VA, Hill AG, Jeffery M, Sarfati D et al. The New Zealand PIPER Project: colorectal cancer survival according to rurality, ethnicity and socioeconomic deprivation-results from a retrospective cohort study. The New Zealand medical Journal. 2018;131(1476):24-39. PubMed | Google Scholar
- Rasouli MA, Moradi G, Roshani D, Nikkhoo B, Ghaderi E, Ghaytasi B. Prognostic factors and survival of colorectal cancer in Kurdistan province, Iran: a population-based study. (2009-2014). Medicine 2017 Feb;96(6):e5941. PubMed | Google Scholar
- Chao-Hsien L, Cheng SC, Hong Yi T, Chang SC, Ching CY, Shu Fen W. The risk factors affecting survival in colorectal cancer in Taiwan. Iranian Journal of Public Health. 2018;47(4):519. PubMed | Google Scholar
- Aryaie M, Roshandel G, Semnani S, Asadi-Lari M, Aarabi M, Vakili MA et al. Predictors of colorectal cancer survival in Golestan, Iran: a population-based study. Epidemiology and health. 2013 Jun 20;35:e2013004. PubMed | Google Scholar
- ACS. Understanding your pathology report: invasive adenocarcinoma of the colon. 2017. Accessed April 20 2021.
- Li Z, Wang K, Zhang X, Wen J. Marital status and survival in patients with rectal cancer: a population-based STROBE cohort study. Medicine (Baltimore). 2018 May;97(18):e0637. PubMed | Google Scholar
- Li Q, Gan L, Liang L, Li X, Cai S. The influence of marital status on stage at diagnosis and survival of patients with colorectal cancer. Oncotarget. 2015;6(9):7339. PubMed | Google Scholar
- Bai DS, Chen P, Qian JJ, Jin SJ, Jiang GQ. Effect of marital status on the survival of patients with gallbladder cancer treated with surgical resection: a population-based study. Oncotarget. 2017;8(16):26404-13. PubMed | Google Scholar
- Pule ML, Buckley E, Niyonsenga T, Roder D. The effects of comorbidity on colorectal cancer mortality in an Australian cancer population. Scientific reports. 2019;9(1):8580. PubMed | Google Scholar
- Salako O, Okediji PT, Habeebu MY, Fatiregun OA, Awofeso OM, Okunade KS et al. The pattern of comorbidities in cancer patients in Lagos, South-Western Nigeria. Ecancermedicalscience. 2018 Jun 13;12:843. PubMed | Google Scholar
- Sarfati D, Koczwara B, Jackson C. The impact of comorbidity on cancer and its treatment. CA: a cancer journal for clinicians. 2016;66(4):337-50. PubMed | Google Scholar
- Lee L, Cheung WY, Atkinson E, Krzyzanowska MK. Impact of comorbidity on chemotherapy use and outcomes in solid tumors: a systematic review. Journal of Clinical Oncology. 2011;29(1):106-17. PubMed | Google Scholar
- Schoenberg NE, Kim H, Edwards W, Fleming ST. Burden of common multiple-morbidity constellations on out-of-pocket medical expenditures among older adults. The Gerontologist. 2007;47(4):423-37. PubMed | Google Scholar
- Hart CL, Davey Smith G, Gruer L, Watt GCM. The combined effect of smoking tobacco and drinking alcohol on cause-specific mortality: a 30 year cohort study. BMC Public Health. 2010;10:789. PubMed | Google Scholar
- Kawano Y. Physio-pathological effects of alcohol on the cardiovascular system: its role in hypertension and cardiovascular disease. Hypertension Research. 2010;33(3):181-91. PubMed | Google Scholar
- Bishehsari F, Magno E, Swanson G, Desai V, Voigt RM, Forsyth CB et al. Alcohol and gut-derived inflammation. Alcohol Res. 2017;38(2):163-171. PubMed | Google Scholar
- Malaguarnera G, Giordano M, Nunnari G, Bertino G, Malaguarnera M. Gut microbiota in alcoholic liver disease: pathogenetic role and therapeutic perspectives. World Journal of Gastroenterology: WJG. 2014;20(44):16639. PubMed | Google Scholar
- Parajuli R, Bjerkaas E, Tverdal A, Le Marchand L, Weiderpass E, Gram IT. Cigarette smoking and colorectal cancer mortality among 602,242 Norwegian males and females. Clinical Epidemiology. 2014 Apr 7;6:137-45. PubMed | Google Scholar
- Chao A, Thun MJ, Jacobs EJ, Henley SJ, Rodriguez C, Calle EE. Cigarette smoking and colorectal cancer mortality in the cancer prevention study II. JNCI: Journal of the National Cancer Institute. 2000;92(23):1888-96. PubMed | Google Scholar
- Walter V, Jansen L, Hoffmeister M, Brenner H. Smoking and survival of colorectal cancer patients: systematic review and meta-analysis. Annals of Oncology. 2014;25(8):1517-25. PubMed | Google Scholar
- Li LF, Chan RL, Lu L, Shen J, Zhang L, Wu WK et al. Cigarette smoking and gastrointestinal diseases: the causal relationship and underlying molecular mechanisms (review). International journal of molecular medicine. 2014;34(2):372-80. PubMed | Google Scholar
- Wu WK, Cho CH. The pharmacological actions of nicotine on the gastrointestinal tract. Journal of pharmacological sciences. 2004;94(4):348-58. PubMed | Google Scholar