Sero-prevalence and risk factors for human brucellosis in Marsabit County, Kenya (2014)
Samuel Maina Kahariri, Philip Mwanzia Kitala, Gerald Mwangi Muchemi, Kariuki Njenga, Mark Nanyingi
Corresponding author: Mark Nanyingi, Department of Public Health, Pharmacology and Toxicology, University of Nairobi, Nairobi, Kenya 
Received: 17 Nov 2020 - Accepted: 08 Feb 2021 - Published: 11 Feb 2021
Domain: Epidemiology,Public health
Keywords: Brucellosis, sero-prevalence, risk factors, Marsabit, Kenya
©Samuel Maina Kahariri et al. PAMJ-One Health (ISSN: 2707-2800). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Samuel Maina Kahariri et al. Sero-prevalence and risk factors for human brucellosis in Marsabit County, Kenya (2014). PAMJ-One Health. 2021;4:9. [doi: 10.11604/pamj-oh.2021.4.9.27024]
Available online at: https://www.one-health.panafrican-med-journal.com/content/article/4/9/full
Sero-prevalence and risk factors for human brucellosis in Marsabit County, Kenya (2014)
Samuel Maina Kahariri1,2, Philip Mwanzia Kitala2, Gerald Mwangi Muchemi2, Kariuki Njenga3, Mark Nanyingi2,4,5,&
&Corresponding author
Introduction: brucellosis is among the world's most widespread zoonotic diseases which is recognized as a public health concern in both developed and developing countries. It is endemic in Kenya's pastoral communities where it is associated with significant economic losses due to decreased animal productivity and high burden in humans. The objectives of this study were: i) To estimate the sero-prevalence and determine the risk factors of brucellosis in humans ii) To assess the knowledge attitude and practices (KAP) of a pastoral community in relation to brucellosis transmission and control.
Methods: cross-sectional survey was conducted within the pastoral ecosystem of Marsabit County. A total of 227 households were randomly selected. Blood samples were aseptically drawn from the selected humans and tested for Brucella immunoglobulin G (IgG) antibodies. Questionnaires were administered via personal interviews to the head of each study household to assess risk factors of transmission. Univariate and subsequently multivariate logistic regression analysis were performed examine the factors independently associated with Brucella seropositivity after adjustment for the effects of other explanatory variables.
Results: the individual Brucella sero-prevalence was estimated at 44% (332/755) and that of the household at (73.13%). Although the majority (85.5%) of the respondents had heard of brucellosis, only a few could identify the disease by clinical signs in both humans and animals. The majority (88.5%) engaged in practices that were likely to enhance Brucella transmission and thus spread. Being a male herder increased the risk of infection by almost twice (OR=1.8136) compared to females. People who were either students, farmers, skilled or non-skilled off farm workers were less likely (OR=0.3053, 0.9038, 0.7749 and 0.2010 respectively) to be infected with brucellosis than housewives. Households where milk was boiled before consumption were less likely (OR=0.404) to have a higher rate of brucellosis infection than those who consumed raw milk. Households that used milk from their own animals were much less likely (OR= 0.1754) to have infection than those that use milk from other sources. Households that kept sheep and those that had members who assisted animals during delivery were more likely to have higher rates of brucellosis than those who never kept sheep and those who never assisted in delivery respectively.
Conclusion: brucellosis is endemic in Marsabit County despite the low levels of knowledge and good control practices by the community. Consumption of raw milk and close contact with animal, particularly sheep are the highest risk factors There is a need for implementation of effective prevention strategies and advocacy practices like targeted livestock vaccinations and public education.
Brucellosis is among the world´s most wide spread zoonotic diseases and recognized as a public health concern in both developed and developing countries [1]. Brucellosis remains one of the most common public health and livestock production problems, especially in Kenya´s pastoral communities. In Africa, countries like Uganda and Eritrea have reported 5-48 new cases per million in humans. It is caused by infection with an intracellular gram-negative coccobacilli of the family Brucellaceae and genus Brucella [2] Brucella melitensis and arbutus are the most common cause of brucellosis in humans worldwide. In East Africa about 21,104,976 cases of livestock brucellosis are reported annually. Human infection occur from frequent exposure to infected animals or contaminated animal products [3]. The disease is spread within livestock herds mainly by ingestion or consumption of feed contaminated with Brucella organisms. Humans are infected by transmission through skin openings/cuts, direct or indirect contact with tissues, urine, vaginal discharges, blood, placentas and aborted fetuses of infected animals [4]. Food borne infection may occur following consumption of unpasteurized milk among other dairy products, uncooked meat and its products, poor hygiene and contact with infected animals [5]. In Kenya, the estimated seroprevalence of the disease in Malindi, Kilifi County and Maralal, Samburu County in Kenya were 25% and 27%, respectively [6].
Globally, brucellosis continues to be a major veterinary and public health problem especially where livestock are a major source of livelihood despite the efforts to achieve eradication from a number of developed countries [7]. In Marsabit County in Kenya, livestock contribute immensely to the livelihood of the residents of whom are predominantly pastoralists keeping mainly sheep, goats, cattle and camels. The frequent interactions between humans and livestock increases their susceptibility to zoonotic diseases. The endemicity of brucellosis in Marsabit County can be attributed to expansive livestock herds; uncontrolled livestock movements; inadequate veterinary support services; and vaccines and husbandry practices that increase the risk of infection. The true burden of brucellosis is often underestimated due to misdiagnosis. The aim of this study was to estimate the seroprevalence of human brucellosis, determine the risk factors of transmission and assess the knowledge attitude and practices of communities in control of brucellosis in Marsabit County, Kenya.
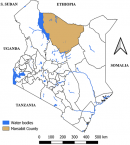
Study sites: the study was conducted in Marsabit County from 2013 to 2014. It is the largest County in Kenya located in the northern part of the country with a size of 66,923 Km2; with a total human population of 291,166 people and density of 4 people per Km2 [8]. It is bordered to the North by Ethiopia, to the West by Turkana County to the South by Samburu County and Isiolo County, and to the East by Wajir County. It borders Lake Turkana on the western side hence its daily average relative humidity is estimated to be 65% all year round [9]. It is an Arid and Semi-Arid land (ASAL) with high temperatures, low rainfall and poor soils with a vegetation cover of shrubs [10]. Pastoral livestock keeping is the main economic activity of the community. It is divided into North Horr, Moyale, Laisamiss and Marsabit sub-counties as indicated in Figure 1.
Study design and sampling: a cross-sectional study was undertaken using a multi-stage sampling method. The sample size was determined based on methods that have a de-fined precision in the entire population, a priori estimate of brucellosis seroprevalence (P) of 15% [6] and confidence level of 95%. The required sample size was estimated to be 196 humans which was increased to 815 after adjusting for clustering using intra-household correlation coefficient of 0.04 [11]. The number of the primary sampling units used was determined using earlier methods by Dohoo et al. [12] as stated in the formula below:
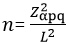
Where: n is the required sample size. Zα is the value of the Z statistic that corresponds to a level of confidence of 95% (1.96). P denotes the prevalence (15%) of brucellosis [6] q=1-p L is the precision of the estimate set at 0.05. With solution:
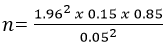
n=196 people. N=196 animals per species. Adjusting for clustering: n'=n(1+ρ(m-1)) Where: n' is the adjusted sample size n is the unadjusted sample size (196); ρ is the intra-household correlation coefficient set at 0.04 [13] and m is the average household size of five. With the solution: n'=196(1+0.04(5-1)); n'=815 human samples (or 271 households); n'=815 animals per species (or 54 herds).
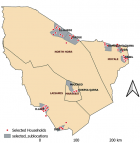
Study sites selection: in January 2014, a systematic multi-stage sampling method was applied where nine sub-locations were randomly selected from the list of all the sub-locations in Marsabit County as per the Kenya national bureau of statistics (KNBS) [14]. The selected sub-locations were; Dabel, Dambala Fachana, El Hadi, Furole, Gurumesa, Illaut, Irir, Majengo, Odda and Rukesa Qarsa. The households were then selected randomly within selected sub-locations. In each selected sub-location, random geographical coordinates were generated in ArcGIS to identify the number of households to be sampled. Sampling was then conducted in one household per geo-code within 5 km radius as shown in Figure 2.
Households identification: a handheld global positioning system (GPS) receiver was used to navigate to the selected geocode. A household to be sampled was then randomly identified using “spin the bottle” method. In this method, a pen or bottle was spun on a surface and the first household towards the direction where the mouth of the bottle or pen tip pointed to was selected.
Sampling of humans: in the selected households, a maximum of 3 persons were sampled. All persons living in the selected household including the herders were listed and the three were randomly selected. The study population were strictly persons aged 5 years and above.
Data collection: data collection was done using a standardized questionnaire in a personal digital assistant (PDA) and geographical coordinates of the sampled households recorded using GPS receivers. The structured questionnaire was administered via personal interviews to the household head. The data collected was on household history of brucellosis, education level, socio-economic status and demographic characteristics. The questionnaire also assessed the knowledge, attitude and practices of the community towards brucellosis.
Blood sample collection: bar coding system was used to label all the cryo-vials and vacutainer tubes that were used in sampling. The sample codes were serialized such that each 5 labels bore the same code, e.g. H08105226 for specimens. Care was taken to ensure that a matching label was fixed to the vacutainer tube and a human tracking sheet. The remaining two labels were transported and delivered to the laboratory with the sera zip-lock bag together with the sample collection form. The identity of the label was counter checked to ensure that it was identical to the forms and sample vials. Five (5) to 7 ml whole blood was drawn via venipuncture of the median cubital vein using a barcoded heparinized Ethylenediaminetetraacetic acid (EDTA) vacutainer tube. The sample was kept at room temperature for 20 min, centrifugation was done for 10 min at 2419.2g. Sera were recovered as aliquots into cryovials and stored at -80°C for shipping and analysis at the Kenya medical research institute (KEMRI)/centers for disease control and prevention (CDC) laboratory in Kisumu.
Laboratory analysis: serological tests on samples were done using the Immuno-Biological Laboratories (IBL)-America IgG ELISA kits according to the manufacturer´s instructions using a 96-well plate pre-coated with inactivated Brucella antigens. Briefly, human sera was diluted at 1:101 with the kit sample diluent then added to microtiter plates pre-coated with Brucella (Brucella abortus strain W99; lysate of a NaCl extract) antigen. All sera and controls were run in duplicates. This was incubated at room temperature for 1hr after which the plates were washed, conjugate then added and incubated for 30min. Following a wash cycle, substrate was added and incubated for 20min. The conjugate-substrate reaction was terminated by the addition of a stop solution. Optical densities (ODs) for the samples were read at 450 nm. To minimize infections strict biosafety measures were observed; all specimens were handled in BSL-2 lab and performed under a safety cabinet. All Brucella positive specimens for culture were handled for identification in BSL-3 lab without retaining aliquots in BSL-2 [15].
Statistical analysis: data were cleaned in excel before being imported to R statistical software version 3.0.2 for analysis. Descriptive statistics were used for explorative analysis including the prevalence estimates which was determined as a fraction of total positives of the entire number of people tested. Generalized linear models (GLM) were used to determine the association between human brucellosis infection and the risk factors. The response variable used was the serological test result. The apparent sero-prevalence was determined at individual and household levels. A seropositive household was defined as any household with at least one person who was seropositive. The independent variables comprised the socio-economic and demographic variables that included age, gender, education level and primary occupation. Animal-related human factors analyzed included livestock ownership, animal contact, working with hides/skins, symptoms experienced within the last year, milk and meat consumption habits and contact with manure and other byproducts. The serological test result, age and sex were collected at individual level while the rest of the human variables were collected at household level. Univariate analysis was first conducted for each potentially explanatory risk factor. The multivariate model included all the significant variables (p-value < 0.05) from the univariate analysis, we sequentially dropped age category, education level, consuming milk from the market, consuming undercooked or uncooked blood, handling hides, milking, slaughtering, cleaning barns and assisting in delivery. The model with the lowest akaike information criterion (AIC) was considered as the most parsimonious. The goodness-of-fit of the final model was tested using residual deviance chi-square (χ2).
Household demographics and characteristics: a total of 755 individuals from the 227 households participated in the study. Slightly more than a half (50.1%) of the respondents were males and the rest were females (Table 1). The average age was 35 years. Sixty-eight (68%) of the respondents had no formal education while only 5.28% of the respondents had formal education beyond the primary level. The primary occupation of 50.3% of the respondents was livestock farming (Table 1). The average household size was five people and 89% of household members consumed milk from their animals. A high proportion (92%) of household members consumed un-boiled milk. A vast majority (87%) of households disposed aborted fetuses and placenta from animals inappropriately by feeding to dogs and leaving it on the pastures. Almost all the surveyed households (88.5%) practiced nomadic pastoralism and the rest, particularly those from the arable part of Marsabit, were agro-pastoralists.
Sero-prevalence: a sero-positivity of 44% (332/755) was detected for brucella antibody in humans. There were variations in seropositivity according to sub-locations ranging from 60% in Furole to 25% in Gurumesa sub-location (Table 2). At household level, 73% (166/227) of the sampled households had at least one member testing positive for brucella antibodies.
Risk factors associated with sero-positivity
Individual factors: univariate analysis showed that handling raw meat and hunting were not significantly associated with seropositivity. Table 3 shows the individual level analysis with outcome variable being seropositivity, the independent variables were gender, age category, education level, primary occupation, consuming packed milk, consuming milk from own animals, consuming milk from the market, consuming undercooked or uncooked blood, handling hides, milking, herding, slaughtering, cleaning barns and assisting in delivery. Students were less likely to be infected with brucellosis compared to people in other occupations (OR=0.3984). Similarly, respondents with formal education were less likely to be infected with brucellosis than those with no education. People who consumed pasteurized milk were less likely to be infected than those who bought their milk from the market. The rest of the variables were risk factors associated with brucellosis infection among the community as shown in the (Table 3). The multivariate model indicated that being a male herder increased the risk of infection by almost twice (OR=1.8136) compared to females. People who were either students, farmers, skilled or non-skilled off farm workers were less likely (OR=0.3053, 0.9038, 0.7749 and 0.2010 respectively) to be infected with brucellosis than housewives (Table 4).
Household factors
Univariate: using the household prevalence as outcome variable, the analysis showed that the significant factors associated (p-value < 0.05) with the household brucellosis sero-positivity included; consuming milk from own animals, consuming milk from vendors shop, feeding aborted materials to dogs, keeping sheep, boiling milk before use and assisting in delivery. There was less likelihood of infection in households that consumed boiled milk compared to raw milk consumption (OR=0.2553, P value < 0.005). Household that kept sheep were twice more likely to have infections of brucellosis compared to those that didn´t . Households where members assisted animals during delivery were 7 times more likely to have a higher rate of brucellosis compared to the unassisted deliveries. Table 5 provides a summary of the univariate analysis at the household level.
Multivariate: the multivariate model showed that households feeding aborted materials to dogs were not significantly associated with brucellosis sero-positivity (p> 0.05) , the variable was therefore dropped from the model. All the other factors that were significant (p < 0.05) were considered as part of the most parsimonious model. Households where milk was boiled before consumption were less likely (OR= 0.404) to have a higher rate of brucellosis infection than those consuming raw milk. Households that consumed milk from their own animals were much less likely (OR=0.1754) to have infections compared to hose sourced elsewhere. Households that kept sheep and those that had members who assisted animals during delivery were more likely to have higher infection rates of brucellosis compared to those that didn´t rear sheep and those who never assisted in delivery respectively (Table 6).
The seroprevalence of human brucellosis was estimated at 44% in Marsabit County, which was higher compared to 5.7% estimated in Kiambu County [16] and relatively significant compared to 32% Kajiado County where nomadic pastoralism is also practiced. Kiambu County´s livestock production system is zero-grazing implying that there is minimal contact of animals between herds and thus the risk of spread of infection is reduced [17]. The overall household seroprevalence was 73%, which is significantly higher compared to the mixed agro-pastoral Kajiado County and the agro-based Kiambu County with 27% and 18% respectively [18]. The high prevalence of brucellosis in the pastoral areas of Marsabit concurs with findings from related studies in Kyrgyzstan which attributed the transmission of human brucellosis to direct contact with animals and also some animal products or indirectly through ingestion of their products [19]. Recent studies in Kenya indicate that nomadic pastoralists are less likely to comply with practices that would minimize brucella infection despite having significant knowledge on risk and transmission of the disease, occupational hazards and cultural practices of consumption of animal products [17].
The community of Marsabit County largely (88.5%) practice pastoralism and are involved in practices that enhance human contact with animals and animal products and thus the high transmission and infection rates of the disease. Consumption of raw milk posed the highest risk among 82.82% of the community members, this has also been reported in other studies that associated transmission of Brucella from animals to human via consumption of raw milk [18]. Lack of education and residing in deeply remote areas was associated with limited knowledge on transmission and control of Brucellosis. We detected an overall individual seroprevalence of 43.97% (332/755) CI (34.24%, 53.70%), which varied between 24% to 57% across all the selected sublocations, an overall household seroprevalence of 73.13% (166/277) was estimated where at least one member was positive for brucellosis and about 54% of the seropositive households had two or three individuals positive. This is relatively higher compared to the household prevalence of brucellosis in humans from Kiambu County which was 5.7% (25/433) and 31.8% (87/274) in Kajiado County [16].
The estimated seroprevalences in this study is considerably higher in comparison to other studies conducted in Kenya, which reported household sero-prevalence of 5.0%, in Kiambu and 5.6% in Kajiado County [20]. This higher prevalence may be attributed to low education levels, nomadic pastoralism that allows the free movement and mixing of stock during grazing and aggregation of animals around water holes that may intensify or amplify infections. This has also been reported in predominant pastoral production systems in Ethiopia and Uganda [21]. The risk factors associated with high seropositivity were advanced age, being a male, consumption of unpasteurized milk, herding, feeding and milking animals and handling hides. These have been related reports Tanzania where the risk of human brucellosis was due to transmission through direct contact with animals or their products or indirectly through consumption of their products [22]. There is need to develop and optimized diagnostic algorithms for brucellosis in humans to overcome the challenges in clinical diagnosis that have been reported in the Mediterranean region, these tests may be used in differential diagnosis with other acute febrile illnesses [23].
Brucellosis sero-prevalence in humans is higher in areas where nomadic pastoralism is practiced. This is due to the high contact between the animals and humans as well as practices like consumption of raw milk. The community knowledge on brucellosis is inadequate to enable the community in implementation of effective control practices that would minimize human exposure and infections. Therefore, there is a great need for behavioral communication change to sensitize the community on desisting from consumption of raw milk and implementation of control strategies like livestock vaccinations and improved hygiene.
This study received financial support as part of a CDC-Kenya project “Epidemiologic and Laboratory Assessment of the Burden of Brucellosis in Kenya” that was funded by Defense Threat Reduction Agency (DTRA) to MKN. MN is a Soulsby One Health fellow, he receives funding from the GCRF, One Health Regional Network for the Horn of Africa (HORN) project, UKRI and BBRSC: BB/P027954/1 and Soulsby Foundation. Funders had no role in the design, analysis and interpretation of the results.
What is known about this topic
- There is widespread occurrence of human and animal brucellosis among pastoral communities in Kenya;
- There are significant differences in knowledge, attitudes and practices in management of human brucellosis between nomadic and non-pastoralist communities in Kenya;
- There is high risk of human brucellosis among pastoralists due to animal interactions.
What this study adds
- Status of knowledge, attitude and practice gaps of human brucellosis in a pastoralist community of Marsabit County in Kenya;
- Understanding of animal and environmental risk factors leading to high burden of human brucellosis in Marsabit County in Kenya.
The authors declare no competing interests.
SMK PMK, MKN and GMM conceived and designed the study, SMK performed fieldwork and the laboratory experiments. SMK, MN and GMM analyzed the data, SMK and MN prepared the manuscript and read and reviewed. All the authors have read and agreed to the final manuscript.
The directorate of veterinary services and CDC Kenya staff offered both logistical and material support. We also appreciate all study participants, community leaders, clinicians and the laboratory technicians from Marsabit County. The first author was granted permission to undertake and publish this work by the directorate of veterinary services.
Table 1: demographic characteristics of human respondents in sampled households, Marsabit County, 2013
Table 2: distribution of sero-prevalence of human brucellosis by sub-location
Table 3: univariate analysis using ELISA test result as the response variable at individual level
Table 4: multivariate analysis at individual level
Table 5: univariate analysis at household level
Table 6: multivariate analysis at household level
Figure 1: map of Kenya showing the geographical location of Marsabit County
Figure 2: map of Marsabit County showing the study sub-locations and households
- Addis M. Public health and economic importance of brucellosis: a review. Public Health. 2015;5(7):68-84. Google Scholar
- Gwida M, Al Dahouk S, Melzer F, R�sler U, Neubauer H, Tomaso H et al. Brucellosis - regionally emerging zoonotic disease? .Croatian medical journal. 2010 Aug;51(4):289-95. PubMed | Google Scholar
- Tsegay A, Tuli G, Kassa T, Kebede N. Seroprevalence and risk factors of brucellosis in abattoir workers at Debre Zeit and Modjo export abattoir, Central Ethiopia. BMC Infect Dis. 2017 Jan 26;17(1):101. PubMed | Google Scholar
- Corbel MJ, Food and agriculture organization of the United Nations, world health organization, world organisation for animal health. Brucellosis in humans and animals. 2006. Google Scholar
- Adesokan HK, Alabi PI, Stack JA, Cadmus SIB. Knowledge and practices related to bovine brucellosis transmission amongst livestock workers in Yewa, south-western Nigeria. J S Afr Vet Assoc. 2013 Mar 6;84(1):E1-5. PubMed | Google Scholar
- Kadohira M, McDermott JJ, Shoukri MM, Kyule MN. Variations in the prevalence of antibody to brucella infection in cattle by farm, area and district in Kenya. Epidemiology and infection. 1997 Feb;118(1):35-41. PubMed | Google Scholar
- McDermott JJ, Arimi SM. Brucellosis in sub-Saharan Africa: epidemiology, control and impact. Veterinary microbiology. 2002 Dec 20;90(1-4):111-34. PubMed | Google Scholar
- Commission for Revenue Allocation. Kenya County Fact Sheets. 2014.
- Kirubi C, Wamicha WN, Laichena JK. The effects of woodfuel consumption in the ASAL areas of Kenya: the case of Marsabit forest. African Journal of ecology. 2000;38(1):47-52. Google Scholar
- Reynolds JF, Virginia RA, Kemp PR, De Soyza AG, Tremmel DC. Impact of drought on desert shrubs: effects of seasonality and degree of resource island development. Ecological Monographs. 1999;69(1):69-106. Google Scholar
- Audigé L. Review of Veterinary Epidemiologic Research by Dohoo, Martin, and Stryhn. The Stata Journal. 2004;4(1):89-92. Google Scholar
- Dohoo IR, Martin SW, Stryhn H. Veterinary epidemiologic research. VER. Inc, Charlottetown, PE, Canada. 2009. Google Scholar
- Juan CC, Pinto D, Salas LA, Camacho JC, Rondón M. and Quintero J. A cluster-randomized controlled trial of handrubs for prevention of infectious diseases among children in Colombia. 31, pp.476-484. Revista Panamericana de Salud P�blica. 2012 Jun;31(6):476-84.. PubMed | Google Scholar
- Kenya National Bureau of Stastics. Kenya Population and Housing Census (2009). Analytical Report on Mortality. 2012. Google Scholar
- Muturi M, Bitek A, Mwatondo A, Osoro E, Marwanga D, Gura Z et al. Risk factors for human brucellosis among a pastoralist community in South-West Kenya, 20 BMC Research Notes. 2018 Dec 5;11(1):865. PubMed | Google Scholar
- Ogola E, Thumbi S, Osoro E, Munyua P, Omulo S, Mbatha P et al. Sero-prevalence of brucellosis in humans and their animals: a linked cross-sectional study in two selected counties in Kenya. Online journal of public health informatics. 2014;6(1). Google Scholar
- Njenga MK, Ogolla E, Thumbi SM, Ngere I, Omulo S, Muturi M et al. Comparison of knowledge, attitude, and practices of animal and human brucellosis between nomadic pastoralists and non-pastoralists in Kenya. BMC public health. 2020 Feb 24;20(1):269. PubMed | Google Scholar
- Osoro EM, Munyua P, Omulo S, Ogola E, Ade F, Mbatha P et al. Strong association between human and animal Brucella seropositivity in a linked study in Kenya, 2012-2013. The American journal of tropical medicine and hygiene. 2015 Aug;93(2):224-231. PubMed | Google Scholar
- Kozukeev TB, Ajellat S, Maes E, Favorov M. Risk factors for brucellosis-leylek cind Kadamjay districts, Batken Oblast, Kyrgyzstan, January-November, 2003. Morbidity and Mortality Weekly Report. MMWR 2006 Apr 28;55(1):31-4. PubMed | Google Scholar
- Osoro EM, Munyua P, Omulo S, Ogola E, Ade F, Mbatha P et al. Strong association between human and animal brucella seropositivity in a linked study in Kenya, 2012-2013. American Journal of Tropical Medicine and Hygiene. 2015 Aug;93(2):224-231. PubMed | Google Scholar
- Magona JW, Walubengo J, Galiwango T, Etoori A. Sero prevalence and potential risk of bovine brucellosis in zerograzing and pastoral dairy systems in Uganda. Tropical animal health and production. 2009 Dec;41(8):1765-71. PubMed | Google Scholar
- John K, Fitzpatrick J, French N, Kazwala R, Kambarage D, Mfinanga GS et al. Quantifying risk factors for human brucellosis in rural northern Tanzania. PLoS One. 2010 Apr 1;5(4):e9968. PubMed | Google Scholar
- Aygen B, Doğanay M, Sümerkan B, Yildiz O, Kayabaş Ü. Clinical manifestations, complications and treatment of brucellosis: a retrospective evaluation of 480 patients. Medecine et maladies infectieuses. 2002;32(9):485-493. Google Scholar