Molecular detection of monkeypox virus in wild rodents and humans in Ibadan, Nigeria; a cross-sectional study
David Sokoyebomi Oluwafemi Abafi, Moses Olubusuyi Adewumi, Olisa Olasukanmi Michael, Grace Sabo Nok Kia, Olanike Kudirat Adeyemo, Selim Adewale Alarape, Clement Adebajo Meseko, Joshua Seyi Oyetunde, Nicodemus Mkpuma, Folajimi Oladimeji Shorunke, Comfort Oluladun Aiki-Raji
Corresponding author: David Sokoyebomi Oluwafemi Abafi, Department of Veterinary Public Health and Preventive Medicine, University of Ibadan, Ibadan, Nigeria 
Received: 07 Nov 2023 - Accepted: 31 Mar 2024 - Published: 17 Apr 2024
Domain: Infectious diseases' epidemiology, Health promotion, Public health
Keywords: Monkeypox, Nigeria, rainforests, human-wildlife, orthopoxvirus DNA polymerase, variola, pustular rash
©David Sokoyebomi Oluwafemi Abafi et al. PAMJ-One Health (ISSN: 2707-2800). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: David Sokoyebomi Oluwafemi Abafi et al. Molecular detection of monkeypox virus in wild rodents and humans in Ibadan, Nigeria; a cross-sectional study. PAMJ-One Health. 2024;13:16. [doi: 10.11604/pamj-oh.2024.13.16.42143]
Available online at: https://www.one-health.panafrican-med-journal.com/content/article/13/16/full
Research 
Molecular detection of monkeypox virus in wild rodents and humans in Ibadan, Nigeria; a cross-sectional study
Molecular detection of monkeypox virus in wild rodents and humans in Ibadan, Nigeria; a cross-sectional study
![]() David Sokoyebomi Oluwafemi Abafi1,&, Moses Olubusuyi Adewumi2,
David Sokoyebomi Oluwafemi Abafi1,&, Moses Olubusuyi Adewumi2, ![]() Olisa Olasukanmi Michael3, Grace Sabo Nok Kia4, Olanike Kudirat Adeyemo1,
Olisa Olasukanmi Michael3, Grace Sabo Nok Kia4, Olanike Kudirat Adeyemo1, ![]() Selim Adewale Alarape1,
Selim Adewale Alarape1, ![]() Clement Adebajo Meseko5, Joshua Seyi Oyetunde6,
Clement Adebajo Meseko5, Joshua Seyi Oyetunde6, ![]() Nicodemus Mkpuma6,
Nicodemus Mkpuma6, ![]() Folajimi Oladimeji Shorunke7, Comfort Oluladun Aiki-Raji8
Folajimi Oladimeji Shorunke7, Comfort Oluladun Aiki-Raji8
&Corresponding author
Introduction: monkeypox, a rare viral disease, was discovered in 1958 in Denmark and sporadically affects humans in Central and West Africa rainforests with a mortality rate of 1-10%. This study in Ibadan, Nigeria, aimed to detect the virus in wild rodents near Human-Wildlife Hotspot interfaces. A molecular detection method was employed to assess the incidence of the monkeypox virus in hunting communities.
Methods: organ and blood samples were collected from wild rodents and hunters, respectively, in Ibadan, Nigeria, to detect the monkeypox virus (MPXV). Deoxyribonucleic acid (DNA) was extracted from the samples and tested using a TaqMan-based assay targeting the Orthopoxvirus DNA polymerase gene to detect a wide range of orthopoxviruses. An additional assay using two MGB Eclipse probes targeting two envelope protein genes (F3L and N3R) was used to detect MPXV specifically. A questionnaire was administered to the hunters to collect demographic data.
Results: TaqMan-based and probe assays (F3L-F290, F3L-R396, N3R-F319, and N3R-R457) failed to detect variola, orthopoxviruses, or non-pox viral rash diseases in wild rodents or hunters in Ibadan, Nigeria. None of the samples tested positive for West African MPXV strains. However, 41% of hunters reported MPXV infection in the past based on clinical symptoms.
Conclusion: monkeypox virus-specific primers and gene proteins failed to detect MPXV in wild animals and hunters in Nigeria. Individuals should be informed about MPXV and report any fever and widespread pustular rash with smaller lesions, especially within 10-14 days of contact with wild animals such as African giant rats, squirrels, and monkeys.
Monkeypox, a sylvatic zoonosis prevalent in Central and West African rainforests, is caused by the monkeypox virus (MPXV), a relative of smallpox within the orthopoxvirus family [1]. Native African rodents are believed to be potential reservoirs. Transmission occurs through direct contact with infected animals during hunting and bush meat consumption. The illness is self-limiting, typically resolved within 21 days, with severe cases more common in children [1]. The case fatality rate ranges from 1-10 percent. Unlike smallpox, there is no specific therapy or vaccine for MPXV. First identified in captive monkeys in 1958, MPXV remains a significant human infection, challenging eradication efforts due to its wide host range [2]. Serological investigations reveal that the monkeypox virus (MPXV) has infected several species in their natural habitats, including rats, squirrels, and non-human primates [3]. After being brought to light in 1997, human monkeypox made a comeback in May 2003 in the central United States when people developed a fever and rash after encountering prairie dogs kept as pets. With 81 cases (40 percent of which were verified by tests), this outbreak was the first in the western hemisphere to affect humans with monkeypox [4]. After an investigation, the source was found to be an international shipment of about 800 small animals from Ghana to Texas, where they were finally distributed through Midwest-based prairie dog sales [5].
In the Democratic Republic of the Congo (formerly Zaire), where smallpox had been declared extinct in 1968, a 9-year-old child was diagnosed with human monkeypox in 1970 [6]. After smallpox was eradicated, monkeypox was discovered. It mostly affects rural, rainforest areas of Central and Western Africa, especially the Congo Basin. In addition to the Democratic Republic of the Congo, it is also found in Cameroon, Gambon, Liberia, Nigeria, Sierra Leone, Congo Brazzaville, Côte d'Ivoire, and South Sudan [7]. The monkeypox virus is divided into two genetic clades: the West African clade, which is less virulent and has a case fatality rate of less than 1%, and the Central African clade, which is more virulent and can cause up to 11% of cases [7]. Human-to-human transmission was historically confined to the Congo Basin clade, linked with severe illness and higher fatality rates compared to the West African clade. In September 2017, Nigeria's Bayelsa state reported its first monkeypox cases, sparking an epidemic across seven states, primarily affecting individuals over 20 [8]. By 2018, Nigeria recorded 45 confirmed and 114 suspected cases, resulting in one fatality in an untreated HIV patient. The Nigeria Centre for Disease Control led containment efforts, treating patients in designated facilities [9].
Infection primarily occurs through contact with infected animals or their fluids, with undercooked meat consumption posing a potential risk. Symptoms mirror smallpox, evolving over 2 to 5 weeks post-exposure [10]. Monkeypox transmission occurs through handling bush meat, animal contact, bodily fluids, or contaminated objects, with rodents serving as primary carriers. Diagnosis involves testing lesion DNA resembling chickenpox [11]. Prevention involves smallpox vaccination, while Cidofovir is a potential therapy. Mortality stands at 10%. Differential diagnoses include smallpox, chickenpox, and various skin infections. Unlike monkeypox, smallpox lacks significant lymphadenopathy, and chickenpox rash mainly appears on the trunk [12,13]. For this research, our primary objectives were twofold: firstly, to ascertain the incidence of the monkeypox virus within wild rodent populations and among the hunting community, and secondly, to identify potential risk factors associated with outbreaks, informing public health interventions and future research directions.
Study design: we carried out a cross-sectional study involving wild rodents: African Giant rats (Cricetomys gambianus,Tamias sibiricus) Squirrels (Tamias sibiricus)] and human subjects within purposive selected study sites in southwestern Nigeria (Ibadan, Oyo State).
Study locations: this study was carried out in six (6) local governments of Ibadan: Akinyele, Egbeda, Ido, Lagelu, Ona Ara, and Oluyole. Ibadan is at longitude 7° 2´ and 7° 40´ E and latitudes 3° 35´ and 4° 10´ N. It is the largest indigenous city in tropical Africa and the capital of Oyo State, Nigeria.
Sampling design: convenience and purposive sampling methods were employed to collect bushmeat samples and administer questionnaires in Ibadan and its suburbs. Tissue samples were obtained from prominent bushmeat markets, while structured questionnaires were administered to consenting sellers and hunters to gather demographic data on monkeypox risk factors. Notable markets included Ido, Tollgate, Odo-Ona Kekere, Omi-Adio, and Akufo bushmeat markets.
Questionnaire administration: structured questionnaires were administered by an interviewer, to consenting hunters and residents within the rural hunting communities in six local governments of Ibadan: Akinyele, Egbeda, Ido, Lagelu, Ona Ara, and Oluyole in Ibadan, where the incidence of monkeypox had been previously reported.
Study populations
Humans: we identified participants (20-50 years old) from hunters and bushmeat vendors in six Ibadan localities in cooperation with local leaders. Convenience sampling was used to approach people during community gatherings. After being informed about the study, the possible dangers of eating wild rats, and the advantages of medical outreach, participants filled out questionnaires. Both written and verbal consent were received. Community health concerns were addressed by providing on-site medical exams and deworming medication to encourage participation.
Animals
Inclusion criteria: wild rodents including squirrels, African giant rats, Gambian rats, and elephant shrews.
Exclusion criteria: wild rodents within the inclusion criteria that had been treated with any medication for at least two weeks before slaughter.
Sample size determination: based on the formula, described by Thrusfield [14]:
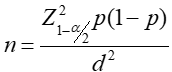
The sample size of bushmeat sellers and hunters was 107. our goal was to achieve a 95% confidence level with a ±5% margin of error in estimating the prevalence of monkeypox among bushmeat sellers and hunters in Ibadan for a statistically reliable estimate. Assumptions included the sample of 107 individuals accurately representing the target population, an expected 7.5% prevalence based on previous studies, and independence among individuals in the sample, ensuring one's monkeypox status does not influence another's.
Sample size: liver, kidney, heart, and lung samples were obtained from 107 wild rodents, while blood samples were obtained from fifty bushmeat sellers and hunters. The sampling technique was based on convenience, determined by the availability of the wild rodents and the hunters´ willingness to be sampled.
Sample collection procedure: tissue samples (brain, lung, liver, spleen, kidney, and ovary/testis) from dead rodents were collected, either freshly killed or euthanized by hunters. Blood samples, collected with anticoagulants (EDTA), and tissue samples in sterile saline water were preserved on ice packs. Samples were tightly sealed in leak-proof containers and transported in plastic bags on ice packs in a laboratory flask. Blood samples from bushmeat sellers and hunters were collected via venepuncture, stored without anticoagulants, labeled correctly, and transported on ice packs to extract serum in the laboratory.
Deoxyribonucleic acid extraction protocol: viral DNA extraction from tissue samples utilized the Qiagen QIAamp™ DNA Mini kit according to the manufacturer's protocol. Initially, tissue pieces were incubated in Buffer ATL and Proteinase K at 56°C for lysis, followed by the addition of Buffer AL and incubation at 70°C. Ethanol was added, and the mixture was transferred onto a QIAamp Mini spin column and centrifuged. The flow-through was discarded, and the column was washed with Buffer AW1 and Buffer AW2. After centrifugation, the column was washed again to eliminate any buffer residue. Finally, Buffer AE was added to elute the DNA, which was collected by centrifugation. This elution step was repeated to enhance DNA yield. The eluted DNA was stored at -20°C for further analysis. This method ensures efficient isolation of viral DNA from tissue samples, employing a series of lysing, washing, and eluting steps to purify the DNA for downstream molecular analyses.
Data analysis: data collected from questionnaires were analyzed using SPSS version 23.0 software. Descriptive statistics, including frequency, percentages, mean, and standard deviation, were employed to summarize the data. To assess the association between the years of experience of hunters and positive monkeypox infections among them, the Chi-square test was employed as a statistical measure. This analysis enables the exploration of potential correlations between the duration of hunting experience and the occurrence of monkeypox infections among the studied population of hunters.
Ethical approval: ethical approval (UI-ACUREC/20/022) was sought from the University of Ibadan - Animal Care and Use in Research Ethical Committee (UI-ACUREC) before the commencement of the project. Ethical approval (UI/EC/20/0096) with registration number: NHREC/05/01/2008a, was also sought for the human samples from the University College Hospital, Ibadan (HREC), and both ethical committees issued certificates to this effect.
Incidence of monkeypox virus: the incidence of the monkeypox virus among hunters in Ibadan, Nigeria (Table 1) is representative of a population that is primarily male (98.0%) and has a diverse age range. Most hunters were married (62.0%) and between the ages of 30 and 49 (80.0%). The primary level had the highest educational achievement (66.0%). Interestingly, 60.0% of hunters said they have been hunting for five to ten years. The aforementioned demographic profile raises concerns about the possibility of male experienced hunters spreading the monkeypox virus, underscoring the need for focused preventative efforts and surveillance tactics in impacted areas.
Association between socio-demographic characteristics and monkeypox infection: in Table 2, while males constituted a significant portion of reporting symptoms, no significant link was observed between sex and monkeypox infection (p = 0.235). Similarly, marital status did not significantly correlate with infection (p = 0.242). Age also showed no significant association (p > 0.05). However, notable geographical disparities were evident, with Lagelu LGA displaying a higher incidence. Interestingly, a trend emerged with education level, as all tertiary-educated individuals reported symptoms. Longer hunting experience showed a slight association with infection (p = 0.841), while consumption of dead animals in forests exhibited a marginally significant link (p = 0.070), suggesting potential risk factors in certain socio-demographic groups.
Result of the PCR tests: Table 3 shows that the real-time PCR tests in Oyo state failed to detect the monkeypox virus (MPXV) in both humans and animals. Negative results indicated MPXV absence. Positive controls had consistent CT values, except for two samples with CT values of 13.24 and 23.01, lacking the typical sigmoid curve. This raised concerns about sample integrity. Overall, the study showed a 0% detection rate of MPXV in tested wild rodent and human samples.
Epidemiological findings and patterns of monkeypox infection: this study, involving 107 rodents and 50 human samples, found no evidence of monkeypox virus. However, questionnaire responses suggested some respondents experienced pox-like rashes after handling and consuming wild animals like monkeys and squirrels, subsequently clearing with local herb use. This hints at potential monkeypox infection, possibly self-limiting due to the West African clade of the virus. Consistent with previous findings, our study noted a 98% male predominance among those exposed to monkeypox, with cases concentrated in individuals aged 30-49 [15].
Comparative analysis of monkeypox infection factors and laboratory findings: our study contrasts with a 2022 study that found no link between marital status, education level, and monkeypox infection [16]. We observed potential correlations, with higher reported symptoms concentrated among married individuals and those with primary education. Additionally, individuals with 5-10 years of hunting experience displayed a higher prevalence of reported symptoms. While bushmeat consumption didn't show a clear association, the interplay of factors like sex, age, and location suggests a complex web influencing monkeypox transmission dynamic. Table 3 details tissue samples from African Giant Rats, squirrels, and blood samples from consenting hunters across six Ibadan local governments. Figure 1 depicts the real-time PCR curve, highlighting the transition from insignificant to detectable fluorescence. Notably, despite no reported monkeypox cases in Nigeria from October 2020 to July 2021, possibly due to reallocated surveillance resources, further testing revealed no MPXV in the remaining samples [17,18].
Epidemiological disparities, surveillance challenges, and global implications of monkeypox transmission: in contrast to our laboratory findings, the Nigeria Center for Disease Control (NCDC) reported 59 probable monkeypox cases in 2021, with 15 verified cases [17,18]. This contrasts with 40.8% of questionnaire respondents claiming similar symptoms. Surveillance challenges in endemic areas include poor infrastructure, scarce resources, and clinical difficulties in recognizing monkeypox illness. Notably, reported and confirmed cases are more prevalent in Nigeria's southern geopolitical zones [19].
There is a dearth of information in the literature regarding the patterns of the monkeypox epidemic, which prevents statistical analysis and results in general conclusions and recommendations. The actual caseload is probably underestimated when patients are excluded from traditional healers and private clinics [20]. Out of Africa, MPXV outbreaks are uncommon and have unclear causes. Although statistically insignificant (P < 0.05), questionnaire-based risk factors do show some correlations [21]. Men were, for instance, 1.4 times more likely to be infected, suggesting possible underlying causes. These results are consistent with research that lists immune system weakness or several sexual partners as non-significant risk factors [22]. There are also contradictions in the identification of certain risk variables, such as residing in a rural area. Because of the many and non-statistically significant risk factors found in different research, the overall understanding of monkeypox transmission is still complicated [23]. The reappearance of monkeypox poses a global concern, as demonstrated by its transmission from Nigeria to Israel, Singapore, and the United Kingdom in recent years, as well as through rodent trafficking to the United States in 2003 [24]. There have been reports of virus transmission from person to person in Nigeria and the UK. Coordination between the human and wildlife health sectors, ecological and epidemiological studies, better laboratory diagnostics, and extensive monitoring are all necessary for effective prevention and control efforts [25,26].
Comprehensive public health strategies and One Health approach to monkeypox prevention and control: public health interventions for monkeypox focus on enhancing diagnostic capacity, early detection, and surveillance. Strategies include public awareness and immunization campaigns, vector control, and infection prevention. Isolation, quarantine, and global cooperation are implemented, alongside community engagement to address cultural behaviors [27]. A coordinated approach integrating research, education, vaccination, and vector management is essential. In the One Health framework, data sharing fosters interdisciplinary collaboration and timely reporting, vital for effective surveillance and health system strengthening to mitigate future outbreaks [28].
Study limitations: we faced resistance from local hunters due to deeply held superstitions, hindering interviews and blood sample collection. Additionally, prevalent power outages affected sample viability, necessitating costly re-collection. Furthermore, obtaining suitable positive controls for molecular detection proved arduous, requiring international collaboration with overseas laboratories. These obstacles underscored the complexities of our study, impacting both budgetary allocations and research timelines.
Our study conducted in August 2021 in Oyo state failed to detect MPXV through laboratory testing in both humans and tested animals. However, questionnaire responses indicated potential past infections. Identified risk factors included sex, marital status, age, locality, education level, hunting experience, and consumption of dead forest animals. Although not statistically significant, these factors suggested complex interactions influencing MPXV transmission. Despite surveillance challenges, this research offers valuable insights into MPXV epidemiology in Oyo state, emphasizing the need for continued vigilance and comprehensive data collection for effective prevention strategies.
What is known about this topic
- Monkeypox is a zoonotic infection that sporadically causes human illnesses in the rainforests of Central and West Africa;
- A possible risk factor is consuming undercooked meat from infected animals;
- Monkeypox has a clinical picture that is quite like smallpox, but the lymph node enlargement that occurs early, typically before the commencement of fever, distinguishes MPXV from smallpox.
What this study adds
- While real-time PCR testing did not detect MPXV in either wild rodents or human participants, this absence does not necessarily negate its potential presence in the region. Further studies employing larger sample sizes and potentially different detection methods may be necessary for a definitive assessment;
- Our analysis of self-reported symptoms alongside demographics revealed potential risk factors (like sex, age, location, and hunting practices which suggest a complex transmission dynamic for monkeypox) that warrant further investigation.
The authors declare no competing interests.
David Sokoyebomi Oluwafemi Abafi conceptualized the study, drafted the manuscript, collected the data, and performed the statistical analysis. David Sokoyebomi Oluwafemi Abafi, Olisa Olasukanmi Michael, Joshua Seyi Oyetunde (of blessed memory), Nicodemus Mkpuma carried out DNA extraction and molecular detection using Real Time PCR, this process was supervised by Olanike Kudirat Adeyemo, Olisa Olasukanmi Michael and Clement Adebajo Meseko. Grace Sabo Nok Kia and Folajimi Oladimeji Shorunke revised the manuscript critically for important intellectual content, Folajimi Oladimeji Shorunke also helped in conforming the manuscript to PAMJ format. All the authors listed read and approved the final manuscript.
We extend our heartfelt appreciation to the entire staff of the Biorepository and Clinical Virology Laboratory, as well as the dedicated team at the Animal Influenza Division, National Veterinary Research Institute, Vom, for their invaluable support and contributions to this research endeavor. We would also like to express our gratitude to Pajoor for his assistance as an interpreter for the rural settlers. Furthermore, we are deeply thankful for the unwavering support and significant contributions from the staff of the Department of Virology in the Faculty of Veterinary Medicine, University of Ibadan. Their expertise and dedication have been instrumental in the successful completion of this research.
Table 1: socio-demographic characteristics of respondents
Table 2: association between socio-demographic characteristics and suspected monkeypox infection
Table 3: tissue samples and the threshold cycle (CT) values
Figure 1: real-time PCR for the tissue samples comprising liver, heart, lungs, and kidney harvested from the wild rodents, as well as blood samples taken from consenting bushmeat sellers and hunters
- World Health Organization. Weekly Bulletin on Outbreaks and Other Emergencies, Week 18 2017. Accessed 6 Nov 2021.
- Adeola F, Fasuyi T, Bakare RA. Re-emergence of monkeypox in Nigeria: a cause for concern and public enlightenment. Afr J Clin Exp Microbiol. 2018 Aug 10;19(4):307-13. Google Scholar
- El Eid R, Allaw F, Haddad SF, Kanj SS. Human monkeypox: a review of the literature. PLOS Pathogens. 2022 Sep 22;18(9):e1010768. PubMed | Google Scholar
- Rimoin AW, Mulembakani PM, Johnston SC, Lloyd Smith JO, Kisalu NK, Kinkela TL et al. Major increase in human monkeypox incidence 30 years after smallpox vaccination campaigns cease in the Democratic Republic of Congo. Proc Natl Acad Sci U S A. 2010;107(37):16262-16267. PubMed | Google Scholar
- Hoff NA, Morier DS, Kisalu NK, Johnston SC, Doshi RH, Hensley LE et al. Varicella Coinfection in Patients with Active monkeypox in the Democratic Republic of the Congo. EcoHealth. 2017 Sep;14(3):564-74. PubMed | Google Scholar
- Bunge EM, Hoet B, Chen L, Lienert F, Weidenthaler H, Baer LR et al. The changing epidemiology of human monkeypox-A potential threat? A systematic review. PLoS Negl Trop Dis. 2022 Feb 11;16(2):e0010141. PubMed | Google Scholar
- Beer EM, Rao VB. A systematic review of the epidemiology of human monkeypox outbreaks and implications for outbreak strategy. PLoS Negl Trop Dis. 2019;13(10): e0007791. PubMed | Google Scholar
- Hutin YJ, Williams RJ, Malfait P, Pebody R, Loparev VN, Ropp SL et al. Outbreak of human monkeypox, Democratic Republic of Congo, 1996 to 1997. Emerg Infect Dis. 2001;7(3):434-438. PubMed | Google Scholar
- Dan-Nwafor C, Ochu CL, Elimian K, Oladejo J, Ilori E, Umeokonkwo C et al. Nigeria´s public health response to the COVID-19 pandemic: January to May 2020. J Glob Health. 2020 Dec;10(2):020399. PubMed | Google Scholar
- European Centre for Disease Prevention and Control. Factsheet for health professionals on monkeypox. 2022. Accessed 6 Nov 2021.
- MODISH PROJECT. Transmission of monkeypox Virus on Animal and Humans. Modish Project. 2018. Accessed Nov 6 2021.
- Burrell CJ, Howard CR, Murphy FA. Laboratory Diagnosis of Virus Diseases. Fenner and White´s Medical Virology. 2017;135-54. Google Scholar
- Jayswal S, Kakadiya J. A narrative review of pox: smallpox vs monkeypox. Egypt J Intern Med. 2022;34(1):90. PubMed | Google Scholar
- Omair A. Sample size estimation and sampling techniques for selecting a representative sample. J Health Spec. 2014;2(4):142. Google Scholar
- Philpott D, Hughes CM, Alroy KA, Kerins JL, Pavlick J, Asbel L et al. Epidemiologic and Clinical Characteristics of monkeypox Cases - United States, May 17-July 22, 2022. 2022:71(32);1018-1022. PubMed | Google Scholar
- Bunge EM, Hoet B, Chen L, Lienert F, Weidenthaler H, Baer LRet al. The changing epidemiology of human monkeypox-A potential threat: A systematic review. PLOS Neglected Tropical Diseases. 2022:16(2);e0010141. PubMed | Google Scholar
- Nigeria Centre for Disease Control (NCDC). First Case of Coronavirus Disease Confirmed in Nigeria. 2021. Accessed Jul 25, 2020.
- Nigeria Centre for Disease Control (NCDC). An Update of monkeypox Outbreak in Nigeria for Week 9. 20. Accessed 11 May 2021.
- Chieloka OS, Bammani IM, Amao LK. Descriptive epidemiology of the burden of human monkeypox in Nigeria: a retrospective review 2017-2021. Pan African Medical Journal One Health. 2022 May 1;8. Google Scholar
- Kang Y, Yu Y, Xu S. Human monkeypox infection threat: A comprehensive overview. PLoS Negl Trop Dis. 2023;17(4): e0011246. PubMed | Google Scholar
- Capobianchi MR, Di Caro A, Piubelli C, Mori A, Bisoffi Z, Castilletti C. Monkeypox 2022 outbreak in non-endemic countries: Open questions relevant for public health, nonpharmacological intervention, and literature review. Front Cell Infect Microbiol. 2022 Sep 20;12:1005955. PubMed | Google Scholar
- Adler H, Gould S, Hine P, Snell LB, Wong W, Houlihan CF et al. Clinical features, and management of human monkeypox: a retrospective observational study in the UK. Lancet Infect Dis. 2022 Aug;22(8):1153-1162. PubMed | Google Scholar
- Ellen M, Beer V, Bhargavi R. A systematic review of the epidemiology of human monkeypox outbreaks and implications for outbreak strategy. PLoS Negl Trop Dis. 2019 Oct 16;13(10):e000779. PubMed | Google Scholar
- Alakunle E, Moens U, Nchinda G, Okeke MI. monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses. 2020;12(11):1257. PubMed | Google Scholar
- Erez N, Achdout H, Milrot E, Schwartz Y, Wiener-Well Y, Paran N, et al. Diagnosis of Imported monkeypox, Israel, 2018. Emerging Infectious Diseases. 2019 May 1;25(5):980-3. PubMed | Google Scholar
- Durski KN, McCollum AM, Nakazawa Y, Petersen BW, Reynolds MG, Briand S et al. Emergence of monkeypox - West and Central Africa, 1970-2017. MMWR Morbidity and Mortality Weekly Report. 2018 Mar 16;67(10):306-10. PubMed | Google Scholar
- Government of Canada. Interim guidance on infection prevention and control for suspect, probable or confirmed monkeypox within healthcare settings. 2022. Accessed Jul 25, 2020.
- World Health Organization. WHO´s 7 policy recommendations on building resilient health systems. 2021. Accessed Jul 25, 2020.





