Bread spoilage mitigation with locally sourced Lactobacilli for improved food safety in Kenitra
Abdessamad Ettouil, Khadija Atfaoui, Asmaa Oubihi, Soumaya Choukri, Mohammed Ouhssine
Corresponding author: Abdessamad Ettouil, Natural Resources and Sustainable Development Laboratory, Department of Biology, Faculty of Sciences, IBN Tofail University, Kenitra, Morocco 
Received: 25 Oct 2023 - Accepted: 26 Nov 2023 - Published: 06 Dec 2023
Domain: Bacteriology, Microbiology, Environmental chemistry/biology
Keywords: Alternaria, antifungal activity, Aspergillus, biopreservatives, lactic acid bacteria, local products, Penicillium, probiotics, sourdoughs
©Abdessamad Ettouil et al. PAMJ-One Health (ISSN: 2707-2800). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Abdessamad Ettouil et al. Bread spoilage mitigation with locally sourced Lactobacilli for improved food safety in Kenitra. PAMJ-One Health. 2023;12:17. [doi: 10.11604/pamj-oh.2023.12.17.42044]
Available online at: https://www.one-health.panafrican-med-journal.com/content/article/12/17/full
Research 
Bread spoilage mitigation with locally sourced Lactobacilli for improved food safety in Kenitra
Bread spoilage mitigation with locally sourced Lactobacilli for improved food safety in Kenitra
![]() Abdessamad Ettouil1,&,
Abdessamad Ettouil1,&, ![]() Khadija Atfaoui1,
Khadija Atfaoui1, ![]() Asmaa Oubihi1, Soumaya Choukri1, Mohammed Ouhssine1
Asmaa Oubihi1, Soumaya Choukri1, Mohammed Ouhssine1
&Corresponding author
Introduction: microorganisms have a critical function in determining the acceptability of foodstuffs manufactured by the food industry. In the bakery and patisserie sector, the fight against the proliferation of molds on the surface of products, generally due to poor hygiene practices and noncompliance with good manufacturing practices, remains one of the priorities for guaranteeing public health. Nevertheless, the utilization of lactic acid bacteria in the bioprocessing of bread products is emerging as a significant substitute for conventional processing aids, thanks to their usefulness as biopreervatives, sourdoughs, improvers, enhancers and probiotics.
Methods: with this in mind, our work will be devoted to exploring the antifungal capabilities of lactic acid bacteria strains obtained from various locally preserved products (olives, peppers, onions, carrots, gherkins, quinces) against contaminating fungal flora recovered from breads from different bakeries in the city of Kenitra, spaced 1 km apart. The phenotypic classification of lactic acid bacteria was carried out on the API gallery after a series of purification tests on selective media.
Results: our findings revealed the presence of five fungal species in the bakery products: Aspergillus niger, Alternaria alternata, Penicillium contaminatum, Fusarium oxysporum, and Rhizopus stolonifer. The distribution of these fungi varied based on the sampling location. Three isolates exhibited substantial inhibitory effects on fungal species.
Conclusion: the antifungal activity of different lactic acid bacterial isolates on fungal species, makes them promising candidates for various technological applications. This study contributes to the understanding of the potential use of locally sourced lactic acid bacteria in mitigating bread spoilage and enhancing food safety in Kenitra, Morocco.
The Moroccan bakery-pastry sector is constantly evolving in terms of organization, technology, logistics, know-how, and manufacturing processes while maintaining a high level of professionalism and innovation to keep pace with consumer demands. Nevertheless, industrialists working in this sector suffer from several threats that hamper the development of their businesses, including the problem of hygiene, which consists of the proliferation of microbial strains that intervene in all stages of the bread-making process right through ready-to-eat products [1]. The emergence of fungi and molds is generally encountered in cereal-based products, which represent a propitious environment for growth, in combination with aeration, which ensures the spread of spores in ceilings and work surfaces, utensils, and equipment used. Raw products, ingredients, packaging, and human factors are the main sources of contamination [2]. This phenomenon is manifested by various fungal species (Aspergillus, Cladosporium, Mucor, Penicillium, etc.) that lead to product deterioration and interfere with the manufacturing process, causing damage to sales and threats to human health through mycotoxins and other toxicogenic substances [1,3].
To address this problem, companies are resorting to excessive use of chemical processing aids, which is the subject of debate as to their efficacy and impact on human health [4]. In this respect, lactic acid bacteria are proving to be a major alternative to food additives since they are recognized for their probiotic and antimicrobial properties on a very broad spectrum of microorganisms [5]. However, the use of allochthonous lactic flora in bread-making is widely desired to increase the rheological and taste qualities of products by means of numerous metabolites produced during the fermentation phenomenon, including alongside lactic acid, the synthesis of bioactive peptides, phenols, exopolysaccharides (e.g., glucans and xanthans), amino acid precursors, aromatic compounds (e.g., acetic acids and alcohols), etc. [6]. These create a favorable environment for the feeding of conventional starters and sourdoughs, resulting in products appreciated by consumers with technological and sensory qualities alongside beneficial superior nutritional properties [7]. Designing a sourdough that combines technological, nutritional, and antimicrobial efficacy involves studying the priorities of the bacterial species selected and the sensitivity of each species of fungus isolated. In this context, our aim will be to study the fungi most frequently found in Moroccan bakery and pastry products, harnessing the probiotic potential of indigenous Lactobacilli from Kenitra's local products and assessing their antifungal properties against the contaminating strains.
Study design and rationale: we conducted a cross-sectional observational study to investigate the antifungal capabilities of lactic acid bacteria strains isolated from locally preserved products and their effect on fungal flora in bread products from bakeries across Kenitra, Morocco. This design provides a snapshot of the relationship between these variables and enhances our understanding of local food safety. Data collection spanned from 11/09/2021 to 10/04/2022. We collected samples from 36 bakeries within Kenitra, located approximately 1 kilometer apart. This approach allowed us to assess locally sourced lactic acid bacteria's antifungal properties against specific bread contaminants, informing strategies for improved food safety in the region. In this study, we exclusively focus on the population of Kenitra, aiming to shed light on the prevalence and mitigation of bread spoilage issues within this community. Nevertheless, an ongoing study about the correlation between the prevalence and factors contributing to mold contamination in bread purchased from local bakeries and human health and safety in the city of Kenitra, Morocco is currently under analysis, and statistical methods will be applied to assess the prevalence of mold contamination and its potential impact on public health and safety. To achieve this, a comprehensive survey has been designed and conducted, involving citizens from various neighborhoods of Kenitra. The survey includes questions about the presence of visible mold on bread, storage practices, concerns related to mold consumption, and differences among local bakeries.
Sampling: the strains were isolated from various biotope samples of fermented vegetables obtained from local households and from local markets in the city of Kenitra: olives, peppers, onions, carrots, gherkins, quinces, and homemade sourdough. The preparation of fermented conserves under laboratory conditions was carried out using a screw-capped bottle by mixing 120 g of veggies with 500 ml of brine (salt water) under sterile conditions. The fermentation of household samples continued for an extended period of up to 11 days, during which isolation procedures were performed. Alternatively, sourdough sampling was conducted by dissolving 10 g in 90 mL of peptone water. During the fermentation process, pH and microbiological analysis of samples were performed aseptically every day. A 1 mL inoculum of each preserved product underwent a series of dilutions spanning from 10�1 to 10�9. The samples were stored at a temperature of 4�C until the subsequent day when microbiological analyses were carried out. The experiment was conducted four independent times. The bread samples were collected from local bakeries in 4 urban areas of the city of Kenira, namely, Khabazat, Mimosas, Ouled Oujih and Saknia, with a spacing of approximately 1 km between each. As soon as they were taken out of the bakery, the bread pieces (baguettes, sandwich loaves, etc.) were aseptically placed into sterile plastic bags after the crumbs had been exposed to the air for four to five minutes. The bags were placed in an incubator set to 20�C for a duration of three weeks.
Screening of lactic acid bacteria strains
Counting and isolating lactic acid bacteria: lactic acid bacteria are enumerated as described in previous works [8] on selective media using 1:10 dilutions after a 3 to 4-minute activation step in a boiling water bath at 40�C. Lactobacilli were plated on Man-Rogosa and Sharp agar (M.R.S, pH 6.2±0.2, BIOKAR), M17 agar (pH 6.9±0. 2, BIOKAR) is used for Streptococcus and Lactococcus spp. and Mayeux Sandine Elliker (M.S.E) agar medium (pH 6.9 ± 0.2 BIOKAR) for leuconostocs [9]. Subsequently, incubation at different temperatures (20, 30�C, and 45�C) for 2 to 5 days was carried out [10]. Well-isolated colonies underwent preliminary identification: macroscopic and microscopic observation, Gram staining, catalase and oxidase tests [11]. Selected colonies were subjected to successive subculturing on appropriate selective media. For extended storage, isolates were maintained at -20�C in M.R.S, M17 or M.S.E. broths supplemented with 15% (v/v) glycerol [12].
Selection and identification of the isolates: for our purposes, the preliminary selection of lactic strains for potential use in bread-making is founded on their capacity to thrive in a synthetic medium derived from wheat flour, in addition to their acidifying power. The synthetic medium was formulated with the following ingredients: wheat flour hydrolysate (T45) 10 g/L, sodium acetate 4.5 g/L, meat extract 5 g/L, Tween 80 1.08 g/L, yeast extract 2.50 g/L, ammonium citrate 2 g/L, dipotassium phosphate 2 g/L, magnesium sulfate 0.2 g/L, and manganese sulfate 50 mg/L, to which 1 g/L of each of the following 4 sugars was added: soluble starch, sucrose, glucose and fructose. The pH was adjusted to 7.00±0.2. Inoculation of 1 mL of inocula was carried out in test tubes similar to those used for sugar fermentation tests, each containing 9 ml of synthetic medium with Durham's bell. A positive outcome is indicated by the visual examination of turbidity and/or the bell's displacement. The remaining isolates were subsequently phenotypically characterized down to the genus using Garabal et al. (2008) criteria, followed by phenotypic identification keys and standard taxonomic descriptions, as described by Wood and Holzapfel (1995) [13,14]. Briefly, the pure cultures were evaluated according to their growth performance at various temperatures (10, 15, and 45�C), pH levels (4 and 9.6), ability to withstand varying salt concentrations (6% and 18.5% NaCl), and ability to hydrolyze citrates and esculin in addition to producing gas from glucose [13]. Additionally, the carbohydrate assimilation and fermentation profiles were determined using API.50 .CH strips and API.CHL medium [14]. All isolates assigned to the species level were identified using APILAB_PLUS (Ver.3.33, Bio-Merieux). The lactic acid bacteria (LAB) strains were preserved at -40�C in either MRS or M17 broth supplemented with 10% glycerol.
Screening of spoilage fungi
Isolation and purification of spoilage fungi from collected breads: the spoilage fungi were isolated from incubated bread samples by three-point inoculation of the wash-off pieces onto Potato Dextrose Agar (PDA) media and were placed in incubation at 25�C for a duration of 3 to 7 days, allowing them to proliferate on the surfaces of Petri dishes [15,16]. Isolation of the pure fungal colony was achieved by successive subculturing of spores on the PDA medium. the plates were incubated in the laboratory incubator for a period ranging from 3 to 5 days at 25�C. The spores from pure culture were stored in PDA medium inside test tubes sealed with parafilm and kept at 4�C for potential future use [17].
Identification of spoilage fungi: the phenotypic identification of spoilage fungi down to the genus stage was carried out following the standardized keys procedure outlined by Pitt and Hocking [18] with some modifications [16]. Briefly, characterization was initiated by transferring the pure strains to various culture media according to three-point inoculation. The study of macroscopic characteristics and preservation of fungi was conducted on yeast extract sucrose agar (YES), as well as on PDA. Czapek yeast extract agar (CYA) was used to examine growth at three temperature levels: 15�C, 30�C and 37�C, while creatine sucrose agar (CREA) was used to indicate acid production. The sporulation test was carried out on malt extract agar (MEA) and achieved on V-8 juice agar (V-8 J) (specifically to distinguish Alternaria from other species). All isolates were kept in the incubator for approximately 1-3 weeks in the dark, following the specific requirements of each assay. Hence, the strains were subjected to several tests of macro- and microscopic characteristics, such as colony color, shape and texture, diameters on each agar medium, mycelium appearance and exudate production.
Antifungal activity of laboratory strains: the assessment of the antifungal efficacy of the chosen lactobacilli strains was performed through a well-diffusion assay [19]. The purified isolates were cultured at 30�C for 36 h in M.R.S broth within 250 ml Erlenmeyer flasks sealed with cotton plugs. Following this, aliquots were incubated anaerobically in test tubes for 18 h at 37�C to reach a concentration of 105 cells/ml. Subsequently, the cultures underwent a series of processes, including a 10-min heating step at 80�C, a 3-min sonication at 6�C and then centrifuged for 10 min at 15,000 g afterward filtered through a membrane filte (0.45 μm; Millipore). The resulting cell-free supernatant underwent freeze-drying and then reconstituted in a solution of 10 mM acetic acid, with pH adjustment to 5.0 using 1 M NaOH and 1 M HCll. Simultaneously, inocula of fungal spores (conidia) were prepared and concentrated to 106 cells/ml by growing the spoilage strains on peptone water until sporulation [5]. Man-Rogosa and Sharp agar Petri dishes with 100 μl of a conidial spore solution were prepared with two 5 mm diameter wells. To prevent any infiltration, a small amount of agar was applied. Subsequently, an 80 μL aliquot of the cell-free supernatant was introduced into the wells, and the plates underwent a two-stage incubation process. This involved an initial pre-incubation of 5 hours at room temperature, succeeded by an aerobic incubation at 30�C for 48 hours. The assessment of antifungal properties was based on the following scale: (-): No suppression observed around the colony; (+): Weak suppression with a range of 0.1-1.0 cm; (+): Suppression characterized by a discernible halo measuring 1.1-2.0 cm; (+): Strong suppression with a clear halo spanning 2.1-3.0 cm; (+): Very strong suppression with a clear halo extending beyond 3.0 cm."
Data collection and statistical analysis: in our study, we explored multiple variables related to fungal contamination, lactic acid bacteria characteristics, and citizen demographics. Rigorous data collection protocols were employed to minimize bias, including systematic bakery selection. For quantitative measurements, we used categorical and continuous variables to assess mold contamination, measured antifungal activity through diameter, and employed quantitative scales for survey data. Statistical methods included the use of descriptive statistics to summarize findings and inferential statistics, such as ANOVA and regression analysis, to assess relationships among variables. Data analysis for the survey was facilitated through the use of SPSS software.
Study design and rationale: our study, involving 376 participants from various neighborhoods in Kenitra, Morocco, investigated mold contamination levels in bread purchased from local bakeries situated in four distinct areas of the city: Khabazat, Mimosas, Ouled Oujih, and Saknia. Participants spanned various age groups, including adolescents, adults, and seniors, and had an even distribution across genders. Statistical analysis, including one-way analysis of variance (ANOVA), revealed significant variations in mold prevalence across these different areas. The ANOVA results indicated that mold contamination rates varied between these areas, with Khabazat having a mean contamination of 12.4% ± 0.84%, Mimosas 8.7% ± 0.91%, Ouled Oujih 15.2% ± 1.05%, and Saknia 10.9% ± 0.76%. Furthermore, our analysis uncovered those specific local bakeries within each area had significantly different rates of mold contamination. These statistical findings emphasize the need for targeted interventions and enhanced food safety measures to address the observed disparities and improve public health and safety in Kenitra. For the second stage of this work, we will address several key findings and their implications.
Screening of lactic acid bacteria strains: over 190 isolates from samples from different biotopes were enumerated on the corresponding selective media (Table 1). A priori, isolations and pH monitoring were carried out at different fermentation stages to collect as many species as possible and ensure product stability. Overall, homemade sourdough showed the highest average LAB concentrations (log cfu/ml) on all plating media: 9.1 log cfu/ml in MRS, 8.5 log cfu/ml in M17 and 4.6 log cfu/ml in M.S.E. In most biotopes, pH decreased steadily with microbial growth until constant values were reached (between days 7 and 8), with the exception of peppers and homemade sourdough, where pH was almost stable between days 4 and 6, with maximum growth on days 7 and 6. It is worth mentioning that in the course of our study, we observed a rise in quantities of LAB starting from the first day of sourdough fermentation, in contrast to the fermentation of vegetables.
Selection and identification of the isolates: first, presumptive LAB strains were grown on synthetic media as described below to satisfy the technological criteria of interest for potential sourdough fermentation in bread-making, which are growth and acidification rate [20]. At this stage, only 23 isolates satisfied the differential criteria for LAB and were either bacilli or cocci, motionless, gram(+) and catalase(-). Morphological observation showed that the majority of LAB strains are rod-shaped and categorized within to the Lactobacillus genus; they are especially mesophilic, including Lactobacillus paracasei, Lactobacillus plantarum, and Lactobacillus acidophilus [11,21]. The remaining strains have a cocci shape and may be attributed to either the Pedicoccus or Lactococcus genus. A summary of the characterization of the relevant LAB strains is presented in Table 2.
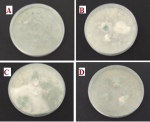
Isolating and purifying spoilage fungi from collected breads: inoculation of the various pieces of bread on PDA medium showed that, in general, each sample was on average contaminated by 3 species of fungi. Spore isolation and purification assays showed that samples from the Saknia area were often contaminated by Penicillium and Rhizopus. The same observation was recorded in the Khabazat area, with a few cases of Aspergillus, while the Mimosas and Ouled Oujih zones were particularly affected by Aspergillus and Fusarium, followed by Alternaria and Mucor sp. (Figure 1). The results showed that 3 groups of fungi are commonly encountered in the majority of cases with a variable distribution depending on the sampling site, namely, Penicillium, Aspergillus and Rhizopus. Otherwise, the occurrence of Alternaria and Fusarium are often observed next to the mucors. The distribution of spoilage fungi according to isolation area and the most representative species for each group are given in Table 3. The phenotypic characterization of the spoilage fungi showed the occurrence of one to two (or even three in rare cases) species representing each fungal family. Penicillium contaminatum, Aspergillus niger, Rhizopus stolonifer, Alternaria alternata and Fusarium oxysporum are the most widespread species. A synopsis of the most frequently encountered strains is shown in the figure below (Figure 2).
Antifungal activity of LAB strains: the antimicrobial proprieties of LAB strains toward spoilage fungi are given in Table 4. Overall, Alternaria alternata displayed sensitivity to all cell-free supernatants of the tested strains. Hence, Rhizopus stolonifer and Penicillium contaminatum have shown resistance against bacterial exudates. Additionally, the strains Bla05, Bl1, and Sbl8 exhibited the highest activities. Similar findings have been reported by numerous studies and have established a link between loss of freshness and fungus proliferation [22-25].
Funding: the present study was conducted and funded by the Natural Resources and Sustainable Development Laboratory, Department of Biology, Faculty of Sciences, Ibn Tofail University, Kenitra, Morocco and received no external funding.
The results obtained in this study are consistent with findings from previous research on traditional Italian fermented legumes compared to control dough and Argentinean fermented kidney beans flours sourdoughs. These studies have emphasized the correlation between the proliferation of biomass, acidification, and proteolytic activities [20,21]. The lactic acid bacteria (LAB) strains isolated in our study, particularly those within the Lactobacillus genus, have demonstrated their potential as good starters for sourdough fermentation. These strains are known for their technological abilities to enhance acidity rapidly, promoting ripening, and improving aroma while overcoming rheological and organoleptic defects due to undesirable flora [22,23]. Furthermore, LAB strains are commonly isolated from vegetables and raw milk and are widely used in dairy products, often in combination with lactobacilli. For example, the combination of Leuconostoc lactis with Lactococcus lactis ssp cremoris in starter bread-making is suitable for aroma production [25]. The inclusion of Pedicoccus alongside Lactobacillus is generally responsible for the sourness of fermented vegetables, with sugar conversion into lactic acid playing a crucial role in extending product shelf life and providing a sour taste [26]. In our study, we also observed the presence of spoilage fungi, including Penicillium, Aspergillus, Rhizopus, Alternaria, and Fusarium, which are known to produce fungal mycotoxins. These mycotoxins are commonly found in food processing and can cause technological and sensory deterioration in the quality of the finished product. They are considered to be a restricting factor for chronic mycotoxicosis. The phenotypic characterization of these spoilage fungi revealed one to two species representing each fungal family, with species like Penicillium contaminatum, Aspergillus niger, Rhizopus stolonifer, Alternaria alternata, and Fusarium oxysporum being the most widespread. These species are associated with various health risks due to the secretion of substances such as aflatoxins, fumonisins, ochratoxins, patulin, trichothecenes, and zearalenone [18]. Significant variations in mold prevalence were observed among different neighborhoods, highlighting the need for targeted interventions to enhance food safety.
Moreover, the screening of indigenous Lactobacilli for potential use in sourdough production and their effectiveness in inhibiting mold contamination, such as Alternaria alternata, demonstrates the promising probiotic potential of these bacteria for enhancing the quality and safety of bakery products. Particularly those from the Lactobacillus genus, were found to be prevalent, with homemade sourdough displaying the highest LAB concentrations. The antifungal activity of LAB strains can be attributed to several factors, including competition due to LAB growth, synergy between secreted compounds like lactic acids and other organic acids leading to a drop in pH, a lack of nutrients, and changes in temperature, creating an inhospitable environment for fungi to thrive [22,24]. Likewise, the production of volatile and bacteriocin components, such as carbon dioxide, ethanol, formic benzoic acids, hydrogen peroxide, diacetyl, acetoin, and nicin, contributes to the antimicrobial properties of LAB strains [21,27]. To summarize, the results of this study highlight the capacity of native Lactobacilli to serve as natural preservatives agents against the proliferation of spoilage molds in baked goods, effectively curbing the growth of spoilage molds and ultimately improving the quality, safety, and overall integrity.
In conclusion, the primary objective of this study was to investigate the spoilage flora in bakery products from the city of Kenitra and explore the probiotic potential of indigenous Lactobacilli sourced from Moroccan products. Our findings revealed the presence of five fungal strains with distribution variations depending on the sampling site. Furthermore, the antifungal activity tests allowed us to propose the formulation of a sourdough containing three specific bacterial species. These results highlight the potential of Lactobacilli as bioconservative agents in mitigating the proliferation of microorganisms, such as spoilage molds, thereby contributing to the enhancement of quality, safety, and wholesomeness in bakery products.
What is known about this topic
- Food safety: the study relates to public health by addressing food safety concerns; controlling fungal contamination in bakery products is essential to prevent potential health hazards associated with consuming spoiled or contaminated foods;
- Probiotic potential: isolating lactic acid bacteria for potential use as probiotics in local Moroccan products is relevant to public health; probiotics have been associated with various health benefits, such as improved digestion and enhanced immunity.
What this study adds
- Localized application: it offers insights into the specific context of Kenitra, Morocco, shedding light on the presence of spoilage flora in bakery products in that area; this localized data can inform food safety practices and product quality improvements in the region;
- Promoting indigenous probiotics: the study highlights the potential use of lactic acid bacteria isolated from local Moroccan products as probiotics; this indigenous approach to probiotics can be valuable for promoting local food industries and offering unique health benefits.
The authors declare no competing interest.
All authors participated in the study including data collection, laboratory work, study design, data analysis, and manuscript preparation. All authors have reviewed and approved the final manuscript.
We express our heartfelt appreciation for the collaborative effort and support that made this research possible. We are grateful to our colleagues and friends for their unwavering encouragement, insightful discussions, and shared enthusiasm throughout the project. Special thanks to the participants whose cooperation and contributions were essential to this study's success.
Table 1: sampling and counting results on different selective media
Table 2: morphological and physiological key characteristics of the presumptive lactic acid bacteria isolates
Table 3: the distribution of spoilage fungi and common species encountered over the areas of sampling
Table 4: antifungal activity of the selected lactic acid bacteria against common spoilage fungi
Figure 1: (A,B,C,D) contaminated bread samples from different areas on Active Pharmaceutical Ingredient (PDA) medium
Keys. A= Khabazat, B= Mimosas, C= Ouled Oujih, D= Saknia
Figure 2: (A,B,C,D,E) microscopic features, and cultural pictures of pure spoilage fungal isolates. Keys. A= Fusarium oxysporum, B= Penicillium contaminatum, C= Alternaria alternata, D= Aspergillus niger, E= Rhizopus stolonifer
- Valle Garcia M, Sonnenstrahl Breg�o A, Parussolo G, Olivier Bernardi A, Stefanello A, Venturini Copetti M. Incidence of spoilage fungi in the air of bakeries with different hygienic status. Int J Food Microbiol. 2019;290:254-261. PubMed | Google Scholar
- Snyder AB, Worobo RW. Fungal Spoilage in Food Processing. J Food Prot. 2018;81(6):1035-1040. PubMed | Google Scholar
- Bernardi AO, Garcia MV, Copetti MV. Food industry spoilage fungi control through facility sanitization. Curr Opin Food Sci. 2019;29:28-34. Google Scholar
- ÇALIŞIR ZE, ÇALIŞKAN D. Food additives and effects on the human health. Journal of Faculty of Pharmacy of Ankara University. 2003;32(3):193-206. Google Scholar
- Schnürer J, Magnusson J. Antifungal lactic acid bacteria as biopreservatives. Trends Food Sci Technol. 2005;16(1-3):70-78. Google Scholar
- Pawlowska AM, Zannini E, Coffey A, Arendt EK. “Green preservatives”: combating fungi in the food and feed industry by applying antifungal lactic acid bacteria. Adv Food Nutr Res. 2012:66:217-38. PubMed | Google Scholar
- Lu Y, Putra SD, Liu S-Q. A novel non-dairy beverage from durian pulp fermented with selected probiotics and yeast. Int J Food Microbiol. 2018;265:1-8. PubMed | Google Scholar
- Khadija A, Omar B, Abdessamad E, Rachid I, Imane O, Hicham H et al. Phenotypic and Genotypic Identification of the Most Acidifiers LAB Strains Isolated from Fermented Food. Biology Bulletin. 2022 Aug;49(4):260-70. Google Scholar
- Harrigan WF. Laboratory Methods in Food Microbiology. Gulf Professional Publishing. 1998. Google Scholar
- Choukri S, Naoufal D, Rachid I, Marieme N, Ettouil A, Atfaoui K et al. Phenotypic identification of five homolactic-type lactic strains, isolated from three different barley-based biotopes. Journal of Xi�an Shiyou University, Natural Science Edition. 2022; volume18.
- Salminen S, Von Wright A, editors. Lactic acid bacteria: microbiological and functional aspects. CRC Press; 2004 Jul 23. Google Scholar
- Elsevier. Laboratory Methods in Microbiology and Molecular Biology. Accessed on August 1, 2023.
- Garabal JI, Rodríguez-Alonso P, Centeno J. Characterization of lactic acid bacteria isolated from raw cows´ milk cheeses currently produced in Galicia (NW Spain). Lwt - Food Sci Technol. 2008;41:1452-1458. Google Scholar
- Wood BJ, Holzapfel WH, editors. The genera of lactic acid bacteria. Springer Science & Business Media; 1992. Google Scholar
- Houbraken J, Visagie CM, Meijer M, Frisvad JC, Busby PE, Pitt JI et al. A taxonomic and phylogenetic revision of Penicillium section Aspergilloides. Stud Mycol. 2014 Jun:78:373-451. PubMed | Google Scholar
- Crous PW, Lombard L, Sandoval-Denis M, Seifert KA, Schroers H-J, Chaerri P et al. Fusarium: more than a node or a foot-shaped basal cell. Stud Mycol. 2021;98:100116. PubMed | Google Scholar
- Dijksterhuis J, Samson RA, editors. Food mycology: a multifaceted approach to fungi and food. CRC Press; 2007 Jun 26. Google Scholar
- Pitt JI, Hocking AD. Fungi and food spoilage. New York: Springer; 2009 Jul 25. Google Scholar
- Magnusson J, Schnürer J. Lactobacillus coryniformis subsp. coryniformis strain Si3 produces a broad-spectrum proteinaceous antifungal compound. Appl Environ Microbiol. 2001 Jan;67(1):1-5. PubMed | Google Scholar
- Sterr Y, Weiss A, Schmidt H. Evaluation of lactic acid bacteria for sourdough fermentation of amaranth. Int J Food Microbiol. 2009;136(1):75-82. PubMed | Google Scholar
- Sáez GD, Hébert EM, Saavedra L, Zárate G. Molecular identification and technological characterization of lactic acid bacteria isolated from fermented kidney beans flours (Phaseolus vulgaris L. and P. coccineus) in northwestern Argentina. Food Res Int. 2017;102:605-615. PubMed | Google Scholar
- Manjarres Melo JJ, �lvarez A, Ramirez C, Bolivar G. Antagonistic Activity of Lactic Acid Bacteria Against Phytopathogenic Fungi Isolated from Cherry Tomato (Solanum lycopersicum var. cerasiforme). Curr Microbiol. 2021;78(4):1399-1408. PubMed | Google Scholar
- El Galiou O, Zantar S, Bakkali M, Laglaoui A, Centeno JA, Carballo J. Chemical and microbiological characteristics of traditional homemade fresh goat cheeses from Northern Morocco. Small Rumin Res. 2015;129:108-113. Google Scholar
- Elbadry M. Preliminary in vitro study on antifungal activity of some local lactobacilli and lactic streptococci isolates. Fayoum J Agric Res Dev. 2008;22(2):129-137. Google Scholar
- Saladino F, Luz C, Manyes L, Fernández-Franzón M, Meca G. In vitro antifungal activity of lactic acid bacteria against mycotoxigenic fungi and their application in loaf bread shelf life improvement. Food Control. 2016;67:273-277. Google Scholar
- Farnworth ER. Kefir a complex probiotic. Food Sci Htmlent Glyphamp Asciiamp Technol Bull Funct Foods. 2005;2(1):1-17. Google Scholar
- Di Cagno R, Coda R, De Angelis M, Gobbetti M. Exploitation of vegetables and fruits through lactic acid fermentation. Food Microbiol. 2013;33(1):1-10. PubMed | Google Scholar