Investigation of furunculosis in cultured catfish
Okoli Solomon Chieloka, Ogugua Akwoba Joseph
Corresponding author: Okoli Solomon Chieloka, Nigerian Field Epidemiology and Laboratory Training Program (NFELTP), Abuja, Nigeria 
Received: 16 Jul 2023 - Accepted: 27 Jul 2023 - Published: 23 Aug 2023
Domain: Epidemiology,Public health,Vector biology
Keywords: Furunculosis, Clarias gariepinus, fluorescent antibody test
©Okoli Solomon Chieloka et al. PAMJ-One Health (ISSN: 2707-2800). This is an Open Access article distributed under the terms of the Creative Commons Attribution International 4.0 License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cite this article: Okoli Solomon Chieloka et al. Investigation of furunculosis in cultured catfish. PAMJ-One Health. 2023;11:15. [doi: 10.11604/pamj-oh.2023.11.15.41095]
Available online at: https://www.one-health.panafrican-med-journal.com/content/article/11/15/full
Investigation of furunculosis in cultured catfish
&Corresponding author
Fish culture accounts for about 40 percent of the total protein intake of households in Nigeria. However, disease outbreaks have negatively affected the growth of the aquaculture subsector in the absence of measures targeted at effective disease prevention and control. Between the 9th and 14th of March 2023, a suspected outbreak of furunculosis in cultured Clarias gariepinus was reported at a backyard fishpond in Ebonyi State, Nigeria. Clinical symptoms seen on affected fishes were furuncles on the skin, bleeding sores (ulcers) on the dorsal, anal fins, mouth of the fish, and exophthalmos. A retrospective and prospective disease investigation was conducted by reviewing case history, sources of fish, hatchery records, stocking density, water quality, sources of breeding stock and treatment regimen. The samples collected were whole fish (fresh carcasses) packaged in a Giostyle and a sample of pond water for laboratory analysis. Mortality was 2%, P.H of pond water was 7.3 (6.5-9) dissolved oxygen (D.O) in pond water was 8.2 ppm (5-20 ppm). The farmer introduced new stock of catfish into the pond from an adjoining farm without proper quarantine measures. This apparently healthy stock was suspected to be the source of the primary infection. Tissue samples from fish tested positive by fluorescent antibody test (FAT) for Aeromonas species and gave an apple green sheen on microscopy. We recommend that fish farmers should ensure proper quarantine of new stock before introducing the same into their fishponds. Prophylaxis with tetracycline may reduce the chances of disease outbreaks. Ensure pond hygiene and eliminate water lice, which could serve as intermediate hosts for Aeromonas salmonicida in the pond.
Furunculosis is a contagious disease of fresh and seawater fishes worldwide. It is caused by a gram-negative nonmotile rod-shaped bacterium Aeromonas salmonicida [1,2]. The disease is characterized by skin ulcers (furuncles), focal or generalized erythema, exophthalmos, and swollen abdomen. It was first identified in Germany in 1904 when a furuncle-like lesion was seen in affected salmonids [1]. Until the late 1960s Aeromonas species was believed to only cause infection in salmonids [1]. However, a strain of Aeromonas salmonicida is responsible for ulcers in goldfish, Atlantic salmons are the most susceptible [1,3]. Furthermore, Aeromonas hydrophila, caviae and veroni have been recognized as opportunistic pathogens in humans where they cause gastrointestinal infection and septicemia, especially in immunocompromised individuals [4]. There are three subspecies of Aeromonas salmonicida, subspecies salmonicida (typical form) is associated with systemic infection (furunculosis), and atypical ”subspecies Aeromonas salmonicida subspecies achromogenes and masoucida, a heterogenous group responsible for localized skin ulcer disease seen in non-salmonid [5]. However, lesions of typical and atypical strains are not pathognomonic [6], contact of naïve fishes with infected cutaneous lesions is the major route for the horizontal spread of infection, although, carrier status may develop among cultured fishes following an epidemic and may account for the disease burden in naïve stocks via viral shedding through faces [3]. It has been shown that the vertical transmission of the infection is rare [7]. In per acute infection, mortality is sudden with darkening of the skin, while the acute form is characterized by hemorrhagic septicemia, especially in growing fish where mortality occurs in 2-3 days [1]. The sub-acute form seen in adults is characterized by hemorrhagic septicemia, which may include exophthalmos and multifocal hemorrhages in the viscera and muscle [8]. The gills may be pale due to anemia [9], stress, high water temperature, the presence of water lice and contact of captive fishes with wild species are risk factors for the outbreak of furunculosis [10]. There is a paucity of information on the outbreak investigation of furunculosis in cultured catfishes in Nigeria. Therefore, the purpose of this study was to investigate an outbreak of furunculosis in a cultured catfish farm to provide information necessary for public health action
Case definition of suspected cases of furunculosis: fish of any age with furuncles on the skin, bleeding sores from skin and bases of fins, destruction of pectoral fins and oral cavity, paling out of gills, discoloration of the skin, anal bleeding, exophthalmos, and abdominal swelling may be regarded as a suspected case [1,3]
Differential diagnosis of furunculosis: Koi herpes virus disease, viral hemorrhagic septicemia [11].
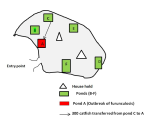
Study area: the outbreak investigation was conducted in a backyard fish farm in Ebonyi State Nigeria (Figure 1) on Latitude. 6°15´N, Longitude. 8°05´E. The farm was an intensive management fish culture involving concrete ponds and a hatchery. Farm holding is located within a cluster of six backyard fish farms with a poor road network accessible only on foot (Figure 2).
Study design: a disease investigation team was constituted comprising the Federal Epidemiology Officer of Ebonyi State, a private veterinary consultant, and a veterinary technician from the Federal Department of Veterinary and Pest Control Services Ebonyi State. A retrospective and prospective disease investigation was conducted by review of case history, sources of fish, hatchery records, stocking density, water quality, sources of breeding stock and treatment regimen.
Samples collected: whole fish (carcass) in a Giostyle maintained at 4-5°C and pond water maintained at environmental temperature were transported to the laboratory for diagnostic tests.
Laboratory diagnosis: tissue samples were subjected to fluorescent antibody test (FAT) as earlier described [12], samples positive for Aeromonas spp. gave apple green fluorescence on microscopy.
Descriptive finding: a suspected case of furunculosis in a backyard catfish farm was reported by a private clinician to the Epidemiology unit of the Federal Department of Veterinary Services Ebonyi State on the 7th of March 2023 and an outbreak investigation was conducted on the 9th to 14th of March 2023. The following observations were made: fishes were active; furuncles were visible on the dorsal (Figure 3) and ventral tail fins (Figure 4). Furthermore, bleeding ulcers were observed on the mouth (Figure 5). The farmer introduced 300 catfish into his pond from an adjoining pond. An outbreak of furunculosis was reported 4 weeks after this action. Mortality was 2% of a new total stock capacity of 2600 catfishes at 15 weeks. The pond water had a pH of 7.3(6.5-9) part per million (ppm), dissolved oxygen, (D.O) was 8.2 ppm (5.0 - 20) ppm, feeding (concentrate) was adequate.
About one million metric tons of fish are produced annually worldwide, most of which are consumed locally, while 10% are exported [13]. As of 2020, an estimated 58.5 million individuals were engaged in aquaculture worldwide, of this 5.6 million (9.6%) are in Africa [14]. In Nigeria, fish culture accounts for 40% of the protein intake of households, estimated at 13.3 kg/person/per year. Although the growth of the aquaculture subsector in Nigeria has been stunted by disease outbreaks, most aquaculture diseases are not investigated, resulting in a loss of opportunities to determine the root causes. Such understanding would be useful in preventing future disease occurrences. The inability of fish farmers to report outbreaks of disease and ineffective disease surveillance has been identified as the bane of aquaculture in Nigeria [15]. The introduction of new stock of catfish into the pond from an adjoining farm without proper quarantine measures could be responsible for the infection recorded in the pond.
This was in tandem with findings by [16,17] who posited that contaminated water with Aeromonas spp., and contact of cultured fishes with wild fish stocks are a risk factor for outbreak of furunculosis. It has been documented that horizontal transmission of infection from infected fishes to naïve stocks can occur by physical contact or by shedding of the bacterium in water [18,19]. Reports have it that the mechanism of horizontal transmission of infection of atypical species of Aeromonas is similar [6,20]. Furthermore [21] identified the presence of Aeromonas salmonicida within ovaries and testis but could not induce intra-ovarian infection. However [22] posited that vertical infection was not a significant route of transmission of infection of A.salmonicida, because cells from infected parents were unlikely to survive at the eyed-egg stage. These findings agreed with [7] who reported that vertical transmission of furunculosis could not demonstrated among the progeny of the broodstocks with previous exposure to infections. The sample was positive for Aeromonas spp. by a fluorescent antibody test. It is worthy of note that Aeromonas spp. are facultative microbes. Therefore, further microbial tests may be necessary for the isolation and characterization of Aeromonas spp. to typical or atypical form [3].
Furunculosis remains a significant disease of cultured fishes in Nigeria. This study identified the introduction of fishes from different production units without proper quarantine measures as being responsible for the outbreak of the disease in the affected farm. We recommend that fish farmers should ensure proper quarantine of new stock before introducing the same into their fishponds. Prophylaxis with tetracycline may reduce the chances of disease outbreaks. Ensure pond hygiene and eliminate water lice, which could serve as intermediate host for Aeromonas Salmonicida in the pond.
The authors declare no competing interests.
Okoli Solomon Chieloka wrote this manuscript Joseph Ogugua Akwoba made corrections and contributed to this Surgeon of Nigeria made corrections and contributed to this manuscript. All the authors have read and agreed to the final manuscript. manuscript. All the authors have read and agreed to the final manuscript.
We acknowledge all staff and all field officers of the Federal and State Department of Veterinary Services Ebonyi who participated in the conduct of the disease investigation.
Figure 1: map of Nigeria showing Ebonyi State
Figure 2: clusters of backyard fish pond where the outbreak of furunculosis occurred
Figure 3: furuncles on the dorsal fins of affected fish
Figure 4: furuncles on the anal fin of affected fish
Figure 5: bleeding ulcers observed around the mouth of affected fish
- Baset A. Status of furunculosis in fish fauna. In: Bacterial Fish Diseases. Elsevier. 2022;257-267. Google Scholar
- Ding W, Miao Q, Bao X, Wang S, Lu J, Lyu M et al. On-site detection of fish furunculosis by combining DNAzyme and carboxyl-functionalized graphene. Front Chem. 2022 Sep 8;10:1015827. PubMed | Google Scholar
- Noga EJ. Fish disease: diagnosis and treatment. John Wiley & Sons. 2010 Jul 7. PubMed | Google Scholar
- Tewari R, Dudeja M, Nandy S, Das AK. Isolation of Aeromonas salmonicida from Human Blood Sample: A Case Report. J Clin Diagn Res. 2014 Feb;8(2):139-40. PubMed | Google Scholar
- Garrity GM, Brenner DJ, Krieg NR, Staley JR, Manual BS. Systematic bacteriology. The Proteobacteria, Part C: The Alpha-, Beta-, Delta-, and Epsilonproteobacteria, Bergey�s Manual Trust, Department of Microbiology and Molecular Genetics. 2005;2. Google Scholar
- Noga EJ, Berkhoff HA. Pathological and microbiological features of Aeromonas salmonicida infection in the American eel (Anguilla rostrata). Fish Pathol. 1990;25(3):127-132. Google Scholar
- Bullock GL, Stuckey HM. Studies on Vertical Transmission of Aeromonas salmonicida. Progress Fish-Cult. 1987;49(4):302-303. PubMed | Google Scholar
- Lee Herman R. Fish Furunculosis 1952-1966. Trans Am Fish Soc. 1968;97(3):221-230. Google Scholar
- Bruno DW. Histopathology of bacterial kidney disease in laboratory infected rainbow trout, Salmo gairdneri Richardson, and Atlantic salmon, Salmo salar L, with reference to naturally infected fish. J Fish Dis. 1986;9(6):523-537. Google Scholar
- Nese L, Enger Ø. Isolation of Aeromonas salmonicida from salmon lice Lepeophtheirus salmonis and marine plankton. Diseases of Aquatic Organisms. 1993;16(1):79-81. Google Scholar
- Science direct. Pathology of Epizootic Ulcerative Syndrome. Accessed July 7, 2023.
- Austin B. Methods for the diagnosis of bacterial fish diseases. Mar Life Sci Technol. 2019;1(1):41-49. Google Scholar
- World fish. World fish in Nigeria. Accessed July 7, 2023.
- FAO. Employment in Fisheries and Aquaculture. Accessed July 7, 2023.
- Chieloka OS, Ameh C, Stephen AA. Evaluation of the avian influenza surveillance system in Enugu State Nigeria, 2015-2017. PAMJ-One Health. 2021 Feb 1;4(6). Google Scholar
- O´Brien D, Mooney J, Ryan D, Powell E, Hiney M, Smith PR et al. Detection of Aeromonas salmonicida, causal agent of furunculosis in salmonid fish, from the tank effluent of hatchery-reared Atlantic salmon smolts. Appl Environ Microbiol. 1994;60(10):3874-3877. PubMed | Google Scholar
- Cipriano RC, Ford LA, Smith DR, Schachte JH, Petrie CJ. Differences in Detection of Aeromonas salmonicida in Covertly Infected Salmonid Fishes by the Stress-Inducible Furunculosis Test and Culture-Based Assays. J Aquat Anim Health. 1997;9(2):108-113. Google Scholar
- Horne JH. Furunculosis in Trout and the Importance of Carriers in the Spread of the Disease. J Hyg (Lond). 1928;28(1):67-78. PubMed | Google Scholar
- Scott M. The Pathogenicity of Aeromonas salmonicida (Griffin) in Sea and Brackish Waters. J Gen Microbiol. 1968;50(2):321-327. PubMed | Google Scholar
- Wiklund T, Dalsgaard I. Occurrence and significance of atypical Aeromonas salmonicida in non-salmonid and salmonid fish species:a review. Dis Aquat Organ. 1998 Feb 26;32(1):49-69. PubMed | Google Scholar
- Rocco C. Cipriano, Graham L. Bullock. Furunculosis And Other Diseases Caused By Aeromonas salmonicida. 2001. Accessed August 7, 2023.
- Skinner FA, Shewan JM. Aquatic microbiology. Academic Press, London 1978;370. Accessed August 7, 2023.